Regenerative Medicine Market Trends and Forecast
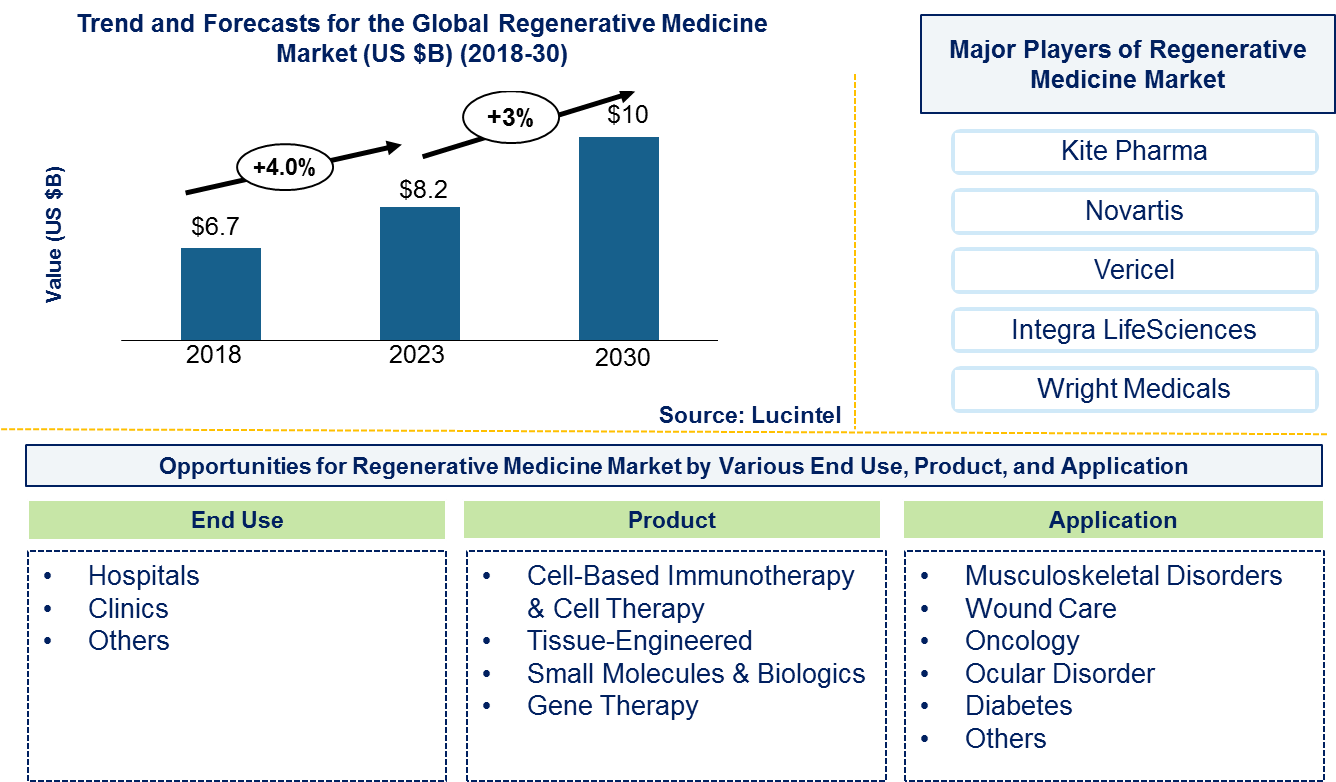
The future of the global regenerative medicine market looks promising with opportunities in musculoskeletal disorders, wound care, oncology, ocular disorders, and diabetes. The global regenerative medicine market is expected to reach an estimated $10 billion by 2030, and it is forecast to grow at a CAGR of 3% from 2023 to 2030. The major drivers for this market are increasing preference of people for personalized medicines, rising investment by governments and private agencies regarding research and development activities, and technological advancements in stem-cell therapies.
The raw materials used in regenerative medicine include biocompatible materials such as scaffolds made from synthetic polymers, natural polymers (e.g., collagen, alginate), and bioceramics (e.g., hydroxyapatite). Growth factors and cytokines derived from recombinant sources or human platelets are crucial for cellular signaling and tissue regeneration. Stem cells, sourced from various tissues (e.g., bone marrow, adipose tissue), are fundamental for their differentiation potential in regenerating tissues. These raw materials play pivotal roles in creating constructs that mimic the extracellular matrix and support cell growth, differentiation, and tissue formation in regenerative medicine applications. The price of regenerative medicine therapies varies significantly depending on factors such as the type of treatment, complexity, and geographic location. Generally, these therapies can be more expensive than traditional treatments due to their innovative nature and personalized approach. Competition in the market is increasing, with companies striving to balance affordability with the high costs associated with research, development, and manufacturing of regenerative medicine products. As technologies advance and economies of scale improve, there is potential for prices to become more competitive in the future.
• Lucintel forecasts that cell-based immunology and cell therapy products will remain the largest segment over the forecast period due to increasing government support in research and development activities related to cell therapy products and establishment of the Cures Act by FDA for streamlining the approval of regenerative medicine.
• Within this market, oncology will remain the largest segment by application over the forecast period due to growing need for cancer-specific regenerative medicine-based therapy options is fueling this market.
• North America will remain the largest region over the forecast period due to presence of advanced technologies and a large number of research institutes involved in the development of novel therapeutics.
• APAC is expected to remain the largest market and is expected to witness highest growth rate during the forecast period due to infrastructure development, and construction of new pipelines.
Country Wise Outlook for the Regenerative Medicine Market
The regenerative medicine market is indeed experiencing significant growth globally, fueled by increased demand from various industries such as healthcare, pharmaceuticals, and biotechnology. Key drivers include advancements in stem cell research, tissue engineering, and gene therapy, which hold promise for treating a wide range of chronic diseases and injuries. Major players in the market are expanding their operations and forming strategic partnerships to strengthen their positions. Below image highlights recent developments by major regenerative medicine producers in key regions: the USA, China, South Korea, Australia, and Japan.
Emerging Trends in the Regenerative Medicine Market
Emerging trends in the regenerative medicine market shaping its future applications and market dynamics:
• Growing Focus on Advanced Therapies: Growing focus on advanced cell therapies, including stem cell treatments and engineered tissues, for addressing complex diseases and injuries.
• Advancements in 3D bioprinting technology: Advancements in 3D bioprinting technology enabling the creation of complex tissues and organs for transplantation and research.
• Integration of Gene Editing Technology: Integration of gene editing tools like CRISPR-Cas9 for modifying cells and tissues to treat genetic disorders.
• Evolving Regulatory Frameworks: Evolving regulatory frameworks to streamline approval processes for regenerative therapies and ensure safety and efficacy.
A total of less than 200 pages has been developed to help in your business decisions. Sample figures with some insights are shown below.
Recent Developments by the Regenerative Medicine Market Suppliers
Recent developments in regenerative medicine by various companies highlight ongoing innovations and advancements across different sectors:
• Advancements in Gene Editing: Continued progress in CRISPR/Cas9 and other gene editing technologies for precise modification of genes, enhancing potential treatments for genetic disorders and cancers.
• Stem Cell Therapies: Increasing focus on stem cell research and therapies, including induced pluripotent stem cells (iPSCs), for regenerating damaged tissues and organs, treating conditions like heart disease and neurological disorders.
• Bioprinting and Organoids: Advancements in 3D bioprinting and the development of organoids-miniature organs grown in vitro from stem cells-for studying diseases and testing drug efficacy.
• Clinical Applications: Growing number of clinical trials and regulatory approvals for regenerative medicine therapies, such as CAR-T cell therapies for cancer and tissue-engineered skin grafts for burns and wounds.
• Refrigerated Warehousing Market: Involves facilities designed to store perishable goods under controlled temperature conditions, ensuring freshness and safety during storage and distribution.
• Regenerative Medicine: Focuses on developing therapies to repair, replace, or regenerate cells, tissues, or organs to restore normal function, often using techniques like stem cell therapy, tissue engineering, and gene editing.
Strategic Growth Opportunities for Regenerative Medicine Market
The regenerative medicine market is very dynamic due to its unique properties of transformative ability to harness biological processes for healing and regeneration, offering personalized treatments tailored to individual patients. Some key strategic growth opportunities for this market include:
Technological Advancements and Innovation
• Chronic diseases and injuries are increasingly influenced by technological advancements in regenerative medicine, including stem cell therapy, biomedical engineering and materials science. The growth of gene editing technologies such as CRISPR-Cas9 could enable personalized therapies and disease modification. Therein lies the opportunity for investment in research and development (R&D) to improve therapeutic efficacy, manufacture scalability, targeted delivery systems- an example is a nano-capsule that enters cancer cells before releasing chemotherapy inside.
Rising Prevalence of Chronic Diseases
• The emergence of chronic conditions like cardiovascular disorders, orthopedic problems, or autoimmune diseases offer significant prospects for regenerative medicine. These treatments seek to repair, replace or regenerate damaged organs or tissues thereby providing alternatives to conventional modes such as surgery or pharmaceuticals. Strategic initiatives in this area include expanding clinical trials through regulatory approval and commercializing products that address areas of unmet need with better patient outcomes.
Personalized Medicine and Biomarker Development
• Growth in the field of regenerative medicine is being driven by the trend towards personalized healthcare models coupled with adoption of biomarker technologies. Consequently it becomes possible to detect diseases early enough employing biomarkers; stratify patients based on specific profiles and monitor their responses to treatment so as they can receive tailored regenerative therapies. This also allows for personalised therapies based on individual patient profiles through exploitation of biomarker data in optimizing treatment protocols; developing companion diagnostics; thereby leading to improved therapeutic outcomes while at the same time reducing healthcare cost.
Regulatory Support and Market Expansion
• Supportive regulatory environments that quicken approvals for regenerative medicine products are necessary for fostering market expansion. Regulatory agencies globally have started viewing these therapies more optimistically hence putting up faster pathways for product development leading into commercialization. Thus, strategic opportunities need navigating complex regulatory landscapes securing marketing authorization from various jurisdictions for novel therapeutics leveraging strategic alliances collaborations partnerships.
Collaboration and Partnerships
• In regard to the commercializing regenerative medicine, collaborations between academic institutions, biotechnology companies and healthcare providers are very important for advancing research, accelerating clinical trials and commercialization. Strategic partnerships offer the necessary expertise, resources and funding that will promote up scaling of production; enable large scale clinical studies to be done as well as meet regulatory requirements. Moreover, they can facilitate introduction of technology into new markets besides aiding penetration into existing ones while stimulating technological innovation in regenerative medicine.
Regenerative Medicine Market Driver and Challenges
Regenerative medicine plays a crucial role across various industries by advancing therapies that harness the body's natural healing processes. It influences sectors such as healthcare, pharmaceuticals, biotechnology, and even cosmetics, offering innovative solutions for treating diseases, injuries, and aging-related conditions.
The factors responsible for driving the regenerative medicine market include:
1. Technological Progress: The capacity of regenerative medicine is broadening due to rapid advances in stem cell studies, tissue engineering and biomaterials. Regenerative medicine is an area that has become attractive as a result of these therapies being offered for conditions that were previously untreatable.
2. Increasing Prevalence of Chronic Diseases: This will highly demand regenerative medicine therapies because cases such as cardiovascular disorders, diabetes, orthopedic injuries are on the rise especially involving chronic diseases. These therapies aim at replacing or repairing damaged tissues and organs thereby providing potential alternatives to traditional treatments like surgery or pharmaceuticals.
3. Aging Population: As people age worldwide, there is increasing need for therapies that address age-related degenerative conditions and improve quality of life. Consequently, regenerative medicine holds hope for solving the diseases associated with aging through initiating tissue regeneration and repair.
4. Personalized Medicine: The shift to personalized medicine has been enhanced by genomic and biomarker developments thus driving targeted regenerative therapeutics. Personalized treatment enhances therapeutic efficacy while minimizing adverse effects by incorporating individual patient characteristics.
Challenges facing the regenerative medicine market are:
1. Complexity of Research and Development: To successfully develop a regenerative therapy requires complex biological processes which necessitates extensive research and development. Some challenges include optimizing cell culture techniques, ensuring clinical trial safety/efficacy as well as scaling up production to meet commercial demand.
2. Regulatory and Ethical Considerations: Regulatory approval processes for regenerative products often face difficulties because they contain new technologies and biological materials. This balance between innovation and patient safety faced by regulatory agencies across the globe leads to different approval timeframes and requirements.
3. High Cost of Treatment: High costs incurred during research, development, manufacturing of drugs, clinical trials make it expensive for many patients who could have otherwise adopted this kind of treatment. In healthcare systems having budget constraints reimbursement issues hinder market penetration resulting in limited access by patients.
4. Long-Term Efficacy and Safety: Ensuring long-term effectiveness along with security in using regenerative medicine therapies is still a major challenge. Tracking and post market surveillance are necessary for managing risks and protecting patients against unforeseen side effects, immune rejection, and tumorigenicity of stem cells.
Innovations in material science and manufacturing processes have spurred a robust demand for regenerative medicine. Unique developments include the creation of biocompatible scaffolds that mimic natural tissues, advanced bioprinting techniques for precise tissue construction, and the integration of nanotechnology to enhance drug delivery and tissue regeneration. These advancements are revolutionizing treatment options by offering personalized solutions for tissue repair, organ regeneration, and therapeutic interventions, driving growth in the regenerative medicine market globally.
Regenerative Medicine Market Suppliers and Their Market Shares
In this globally competitive market, several key players such as Zimmer Biomet, Novartis, Vericel., Integra Life Sciences, Wright Medical etc. dominate the market and contribute to industry’s growth and innovation. These players capture maximum market share. To know the current market share of each of major players contact us.
Companies in the market compete on the basis of product quality offered. Major players in this market focus on expanding their manufacturing facilities, R&D investments, infrastructural development, and leverage integration opportunities across the value chain. With these strategies regenerative medicine companies cater increasing demand, ensure competitive effectiveness, develop innovative products & technologies, reduce production costs, and expand their customer base. Some of the regenerative medicine companies profiled in this report include.
• Zimmer Biomet
• Novartis
• Vericel
• Integra LifeSciences
• Wright Medical
These companies have established themselves as leaders in the regenerative medicine market, with extensive product portfolios, global presence, and strong research and development capabilities. They continually strive to enhance their market positions through strategic partnerships, mergers and acquisitions, and product innovations.
The market share dynamics within the regenerative medicine market are evolving, with the entry of new players and the emergence of innovative regenerative medicine market technologies. Additionally, collaborations between material suppliers, manufacturers, and end-users are fostering technological advancements and expanding market opportunities.
Regenerative Medicine Market by Segment
The regenerative medicine market is seeing significant growth from diverse sectors including healthcare, pharmaceuticals, and biotechnology. This surge is driven by increasing investments in research and development, rising prevalence of chronic diseases, and advancements in technologies like stem cell therapy and tissue engineering.
This regenerative medicine market report provides a comprehensive analysis of the market's current trends, growth drivers, challenges, and future prospects in all major segments like above. It covers various segments, including product, application, and end use. The report offers insights into regional dynamics, highlighting the major markets for regenerative medicine and their growth potentials. The study includes trends and forecast for the global regenerative medicine market by product, application, end use, and region as follows:
By Product [Value ($ Million) analysis from 2018 – 2030]:
• Cell-Based Immunotherapy & Cell Therapy Products
• Tissue-Engineered Products
• Small Molecules and Biologics
• Gene Therapy Products
By Application [Value ($ Million) analysis from r 2018 – 2030]:
• Musculoskeletal Disorders
• Wound Care
• Oncology
• Ocular Disorders
• Diabetes
• Other Applications
By End Use [Value ($ Million) analysis from 2018 – 2030]:
• Hospitals,
• Clinics
• Others
By Region [Value ($ Million) analysis from 2018 – 2030]:
• North America
o United States
o Canada
o Mexico
• Europe
o United Kingdom
o Germany
o France
• Asia Pacific
o China
o India
o Japan
• The Rest of the World
o Brazil
Features of the Regenerative Medicine Market
• Market Size Estimates: Regenerative medicine market size estimation in terms of value ($M).
• Trend and Forecast Analysis: Market trends (2018-2023) and forecast (2024-2030) by various segments and regions.
• Segmentation Analysis: Regenerative medicine market size by various segments, such as end use, product, and application.
• Regional Analysis: Regenerative medicine market breakdown by North America, Europe, Asia Pacific, and the Rest of the World.
• Growth Opportunities: Analysis on growth opportunities in different end use, product, application, and regions for regenerative medicine market.
• Strategic Analysis: This includes M&A and competitive landscape for the regenerative medicine .
• Analysis of competitive intensity of the industry based on Porter’s Five Forces model.
If you are looking to expand your business in global regenerative medicine markets, then contact us. We have done hundreds of strategic consulting projects in market entry, opportunity screening, due diligence, supply chain analysis, M & A, and more.
FAQ
Q1. What is the regenerative medicine market size?
Answer: The global regenerative medicine market is expected to reach an estimated $10 billion by 2030.
Q2. What is the growth forecast for regenerative medicine market?
Answer: The regenerative medicine market is expected to grow at a CAGR of 3% from 2024 to 2030.
Q3. What are the major drivers influencing the growth of the regenerative medicine market?
Answer: The major drivers for this market are increasing preference of people for personalized medicines, rising investment by governments and private agencies regarding research and development activities, and technological advancements in stem-cell therapies.
Q4. What are the major applications or end use industries for regenerative medicine?
Answer: Hospital and clinic are the major end uses for regenerative medicine.
Q5. What are the emerging trends in regenerative medicine market?
Answer: Emerging trends, which have a direct impact on the dynamics of the industry, include Growing focus on advanced cell therapies, advancements in 3D bioprinting technology, integration of gene editing, and evolving regulatory frameworks.
Q6. Who are the key regenerative medicine companies?
Answer: Some of the key regenerative medicine companies are as follows:
• Novartis
• Vericel
• Integra LifeSciences
• Wright Medical
• MiMedx
• Osiris Therapeutics
• Stryker
• Spark Therapeutics
• Zimmer Biomet
Q7.Which regenerative medicine product segment will be the largest in future?
Answer: Lucintel forecasts that cell-based immunology and cell therapy products will remain the largest segment over the forecast period due to increasing government support in research and development activities related to cell therapy products and establishment of the Cures Act by FDA for streamlining the approval of regenerative medicine.
Q8: In regenerative medicine market, which region is expected to be the largest in next 5 years?
Answer: North America will remain the largest region in next 5 years.
Q9. Do we receive customization in this report?
Answer: Yes, Lucintel provides 10% Customization Without any Additional Cost.
This report answers following 11 key questions
Q.1 What are some of the most promising potential, high-growth opportunities for the global regenerative medicine market by product (cell-based immunotherapy & cell therapy products, tissue-engineered products, small molecules and biologics, and gene therapy products), application (musculoskeletal disorders, wound care, oncology, ocular disorders, diabetes, and other applications), and region (North America, Europe, Asia Pacific, and Rest of the World)?
Q. 2 Which segments will grow at a faster pace and why?
Q.3 Which region will grow at a faster pace and why?
Q.4 What are the key factors affecting market dynamics? What are the drivers and challenges of the market?
Q.5 What are the business risks and threats to the market?
Q.6 What are the emerging trends in this market and the reasons behind them?
Q.7 What are some changing demands of customers in the market?
Q.8 What are the new developments in the market? Which companies are leading these developments?
Q.9 Who are the major players in this market? What strategic initiatives are being implemented by key players for business growth?
Q.10 What are some of the competitive products and processes in this market, and how big of a threat do they pose for loss of market share via material or product substitution?
Q.11 What M & A activities did take place in the last five years in this market?
For any questions related to regenerative medicine market or related to regenerative medicine companies, regenerative medicine market size, regenerative medicine market share, regenerative medicine analysis, write Lucintel analyst at email: helpdesk@lucintel.com. We will be glad to get back to you soon.