Report Feature
RT-PCR Kit Market Trends and Forecast
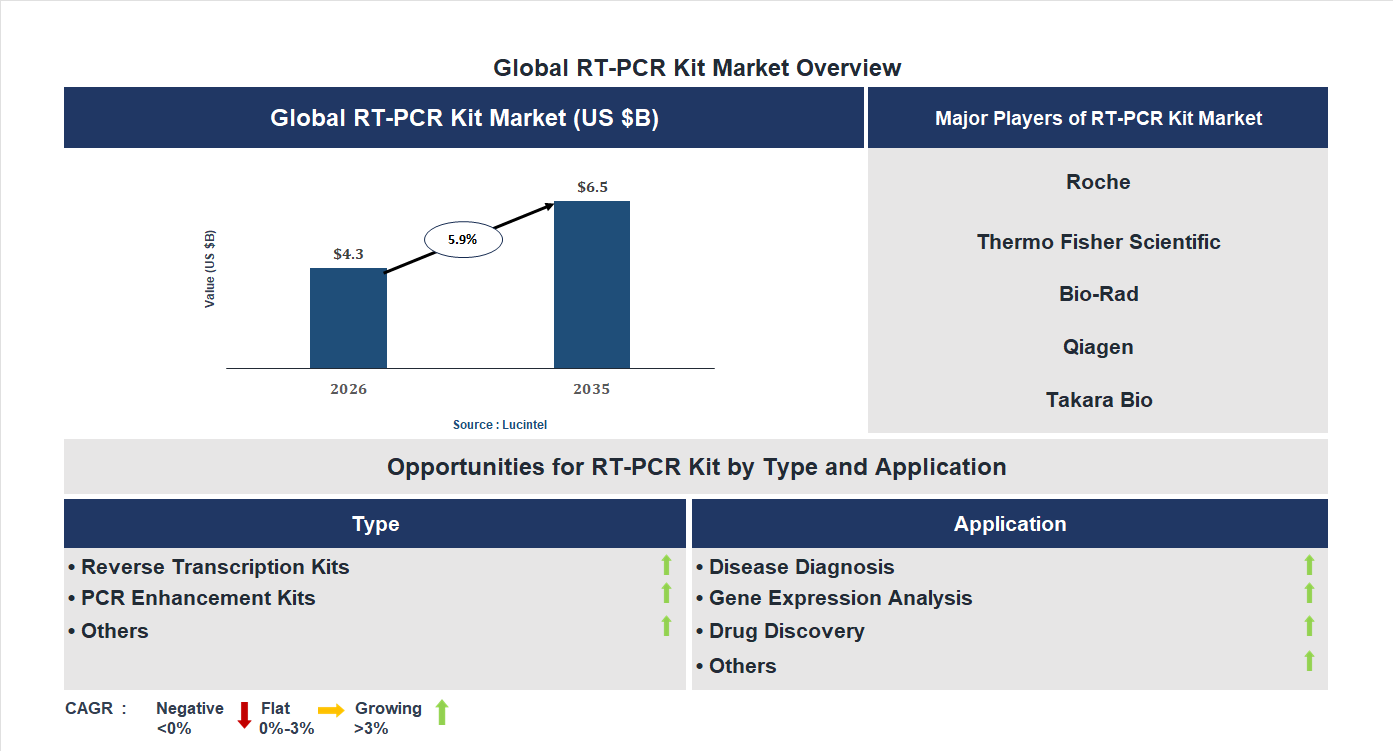
The future of the global RT-PCR kit market looks promising with opportunities in the disease diagnosis, gene expression analysis, and drug discovery markets. The global RT-PCR kit market is expected to reach an estimated $6.5 billion by 2035 with a CAGR of 5.9% from 2026 to 2035. The major drivers for this market are the increasing demand for rapid disease detection, the rising prevalence of infectious diseases, and the growing adoption in clinical diagnostic laboratories.• Lucintel forecasts that, within the type category, reverse transcription kit is expected to witness higher growth over the forecast period.
• Within the application category, disease diagnosis is expected to witness the highest growth.
• In terms of region, APAC is expected to witness the highest growth over the forecast period. Gain valuable insights for your business decisions with our comprehensive 150+ page report. Sample figures with some insights are shown below.
Emerging Trends in the RT-PCR Kit Market
The RT-PCR kit market is experiencing rapid growth driven by advancements in molecular diagnostics, increased demand for accurate testing, and the ongoing need for COVID-19 testing. As the global healthcare landscape evolves, several emerging trends are shaping the future of this market. These trends are influencing product development, market strategies, and healthcare policies, ultimately improving diagnostic accuracy and accessibility. Understanding these key developments is essential for stakeholders aiming to capitalize on opportunities and address challenges in this dynamic environment. The following are five significant trends currently shaping the RT-PCR kit market.• Technological Innovations in RT-PCR Kits: The market is witnessing continuous advancements in RT-PCR technology, including the development of faster, more sensitive, and more specific kits. Innovations such as multiplex RT-PCR, digital PCR, and point-of-care testing are enhancing diagnostic capabilities. These improvements reduce turnaround times, increase accuracy, and enable the detection of multiple pathogens simultaneously. As a result, healthcare providers can deliver quicker and more reliable diagnoses, which is crucial during pandemics and for managing infectious diseases. This trend is driving market growth and expanding application scopes.
• Rising Adoption of COVID-19 Testing Kits: The COVID-19 pandemic has significantly accelerated the demand for RT-PCR testing kits worldwide. Governments and healthcare organizations are prioritizing large-scale testing to control virus spread, leading to increased production and procurement of COVID-19-specific RT-PCR kits. This surge has not only boosted market revenues but also prompted innovations tailored for pandemic response, such as rapid and high-throughput testing solutions. The pandemic has underscored the importance of reliable molecular diagnostics, making COVID-19 testing kits a dominant segment and influencing future market directions.
• Growing Focus on Point-of-Care Testing: There is a notable shift towards developing portable, easy-to-use RT-PCR kits suitable for point-of-care (POC) testing. These kits enable rapid diagnosis outside traditional laboratory settings, such as clinics, remote areas, and field environments. The trend is driven by the need for immediate results, especially in outbreak scenarios and resource-limited settings. POC RT-PCR kits improve healthcare accessibility, reduce diagnostic delays, and facilitate timely treatment decisions. This movement towards decentralization is expected to expand market reach and foster innovation in portable diagnostic technologies.
• Increasing Investment in Research and Development: Market players are investing heavily in R&D to improve RT-PCR kit performance, reduce costs, and expand applications. Innovations include developing kits capable of detecting emerging pathogens, enhancing automation, and integrating with digital health platforms. These investments aim to meet the evolving needs of healthcare providers and patients, ensuring higher accuracy and efficiency. R&D efforts also focus on simplifying procedures to enable broader use by non-specialist personnel. This trend is vital for maintaining competitive advantage and addressing future diagnostic challenges, ultimately driving market expansion.
• Regulatory Approvals and Standardization Efforts: The market is witnessing increased regulatory scrutiny and efforts to standardize RT-PCR testing protocols. Governments and international agencies are establishing guidelines to ensure test reliability, quality, and safety. Obtaining regulatory approvals is becoming more streamlined, encouraging innovation while maintaining standards. These efforts foster market confidence, facilitate international trade, and promote widespread adoption of validated RT-PCR kits. Standardization also helps in comparing test performances and ensuring consistent diagnostic outcomes, which is critical for clinical decision-making and public health strategies. These emerging trends are transforming the RT-PCR kit market by enhancing technological capabilities, increasing testing accessibility, and ensuring quality standards. They are fostering innovation, expanding application areas, and improving diagnostic accuracy, which collectively are reshaping the landscape of molecular diagnostics and positioning the market for sustained growth.

Recent Development in the RT-PCR Kit Market
The RT-PCR kit market is experiencing rapid growth driven by advancements in diagnostic technology, increased demand for accurate testing, and global health challenges. Innovations in assay sensitivity, automation, and supply chain improvements are expanding market reach. Governments and healthcare providers are investing heavily in diagnostic solutions to manage infectious diseases. This evolving landscape presents significant opportunities for manufacturers and stakeholders to enhance product offerings, improve accessibility, and meet the rising demand for reliable testing solutions worldwide.• Growing Demand for COVID-19 Testing: The ongoing pandemic has significantly increased the need for RT-PCR kits, making them essential for accurate detection of SARS-CoV-2. This surge has driven market expansion, prompting manufacturers to innovate faster, improve kit sensitivity, and increase production capacity. The demand is expected to sustain as testing remains critical for controlling outbreaks, influencing global health strategies, and boosting overall market growth.
• Technological Advancements in RT-PCR Assays: Innovations such as multiplex testing, automation, and faster turnaround times are transforming the market. These advancements improve diagnostic accuracy, reduce processing time, and lower costs, making RT-PCR kits more accessible. Enhanced assay sensitivity and specificity are also expanding applications beyond infectious diseases, including oncology and genetic testing, thereby broadening market scope and increasing adoption across various healthcare sectors.
• Increasing Adoption in Point-of-Care Testing: The shift towards decentralized testing is fueling demand for portable, easy-to-use RT-PCR kits suitable for point-of-care settings. These kits enable rapid diagnosis outside traditional laboratories, improving patient management and reducing turnaround times. The development of user-friendly, compact devices is expanding testing capabilities in remote and resource-limited areas, thereby increasing market penetration and supporting healthcare infrastructure development.
• Regulatory Approvals and Standardization: Stringent regulatory frameworks and approval processes are shaping the market by ensuring product quality and safety. Recent approvals of new RT-PCR kits have accelerated market entry and adoption. Standardization efforts are improving test reliability and comparability across regions, fostering trust among healthcare providers and consumers. These developments facilitate international trade, enhance market stability, and encourage innovation within the industry.
• Supply Chain Optimization and Manufacturing Capacity: Enhancements in supply chain logistics and increased manufacturing capacity are addressing global demand fluctuations. Companies are investing in raw material sourcing, automation, and regional manufacturing hubs to ensure timely delivery and reduce shortages. These improvements are critical for maintaining market stability, especially during health crises, and are enabling the industry to meet rising demand efficiently while reducing costs. The recent developments in the RT-PCR kit market, including technological innovations, increased adoption in decentralized testing, regulatory progress, and supply chain improvements, are collectively driving substantial growth. These advancements are enhancing diagnostic accuracy, accessibility, and market stability, ultimately expanding the global reach of RT-PCR testing. As a result, the market is poised for sustained expansion, supporting global health initiatives and improving disease management capabilities worldwide.
Strategic Growth Opportunities in the RT-PCR Kit Market
The RT-PCR kit market is experiencing rapid expansion driven by increasing demand for accurate diagnostic tools, technological advancements, and the need for early disease detection. Growing applications in healthcare, research, and environmental testing are fueling market growth. Strategic collaborations, innovation, and regulatory approvals are further propelling this sector. As the global focus on infectious disease management intensifies, the market presents significant opportunities for manufacturers and stakeholders to innovate and expand their reach across diverse applications.• Increasing Adoption of RT-PCR Kits in Infectious Disease Diagnostics: The rising prevalence of infectious diseases like COVID-19, influenza, and other viral infections has significantly boosted demand for RT-PCR kits. Their high sensitivity and specificity make them essential for early detection and containment efforts. Governments and healthcare providers are investing heavily in diagnostic infrastructure, further expanding the market. Additionally, the need for rapid, reliable testing solutions in clinical and point-of-care settings is driving innovation and adoption of advanced RT-PCR technologies.
• Technological Innovations Enhancing RT-PCR Kit Performance: Advances in molecular biology and biotechnology are leading to the development of more efficient, user-friendly RT-PCR kits. Innovations such as multiplex testing, automation, and miniaturization are improving throughput, reducing turnaround times, and lowering costs. These technological improvements enable broader application in research, clinical diagnostics, and environmental testing. Companies investing in R&D are gaining competitive advantages by offering kits with higher accuracy, faster results, and easier workflows, thus expanding market opportunities.
• Growing Demand for RT-PCR Kits in Research and Development: The expanding research landscape, especially in genomics, personalized medicine, and vaccine development, is fueling demand for RT-PCR kits. Researchers require reliable tools for gene expression analysis, mutation detection, and pathogen identification. The increasing number of research projects and funding initiatives worldwide is driving market growth. Additionally, collaborations between academia and industry are fostering innovation, leading to the development of specialized kits tailored for specific research applications, thereby broadening market scope.
• Rising Use of RT-PCR Kits in Environmental and Food Testing: Environmental monitoring and food safety testing are emerging markets for RT-PCR kits. They are used to detect pathogens, contaminants, and genetically modified organisms efficiently. Stringent regulations and consumer awareness are prompting industries to adopt molecular testing methods for quality control. The portability and rapid results of RT-PCR kits make them suitable for on-site testing, expanding their application beyond healthcare. This diversification is creating new revenue streams and growth avenues for manufacturers.
• Strategic Collaborations and Regulatory Approvals Accelerating Market Growth: Partnerships between biotech firms, diagnostic companies, and research institutions are facilitating product innovation and market penetration. Regulatory approvals and clearances are essential for commercialization, and ongoing efforts to streamline approval processes are boosting market confidence. These collaborations enable access to new markets, enhance product portfolios, and foster technological advancements. As regulatory landscapes evolve, companies that navigate these effectively will capitalize on emerging opportunities, ensuring sustained growth and market leadership. The overall impact of these opportunities is a robust expansion of the RT-PCR kit market, driven by technological progress, increased applications, and strategic collaborations. This growth will enhance diagnostic capabilities, improve healthcare outcomes, and foster innovation across sectors. As demand continues to rise globally, stakeholders are positioned to benefit from new market segments and technological advancements, ensuring sustained market vitality and competitiveness.
RT-PCR Kit Market Driver and Challenges
The RT-PCR kit market is influenced by a variety of technological, economic, and regulatory factors that shape its growth and development. Advances in molecular diagnostics technology, increasing demand for rapid and accurate testing, and supportive government policies are key drivers. Conversely, challenges such as high costs, regulatory hurdles, and supply chain disruptions pose significant obstacles. Understanding these drivers and challenges is essential for stakeholders to navigate the evolving landscape effectively and capitalize on emerging opportunities. The factors responsible for driving the RT-PCR kit market include:-• Technological Advancements: The continuous development of more sensitive, specific, and faster RT-PCR kits has significantly enhanced diagnostic accuracy and efficiency. Innovations such as multiplex testing and automation have streamlined workflows, reducing turnaround times and increasing throughput. These technological improvements meet the rising demand for reliable testing, especially during pandemics, and support the expansion of testing capabilities across healthcare settings. As technology evolves, the market is expected to see increased adoption and new product launches, further fueling growth.
• Rising Prevalence of Infectious Diseases: The global increase in infectious diseases, including COVID-19, influenza, and other viral infections, has created an urgent need for accurate diagnostic tools. RT-PCR kits are considered the gold standard for detecting viral RNA, making them essential in disease management and containment efforts. The growing burden of infectious diseases worldwide, especially in developing regions, drives demand for scalable and reliable testing solutions, thereby expanding the market.
• Government Initiatives and Regulatory Support: Governments worldwide are actively promoting molecular diagnostic testing through funding, subsidies, and regulatory approvals. During health crises like the COVID-19 pandemic, authorities fast-tracked approvals and increased procurement of RT-PCR kits. Such supportive policies and regulatory frameworks facilitate market growth by ensuring product quality, safety, and accessibility. Additionally, ongoing investments in healthcare infrastructure further bolster the adoption of RT-PCR testing.
• Increasing Adoption in Non-Clinical Applications: Beyond clinical diagnostics, RT-PCR kits are increasingly used in research, food safety, and environmental testing. This diversification broadens the market scope and creates new revenue streams. The expanding application base encourages manufacturers to innovate and develop specialized kits, which in turn stimulates market growth. As awareness of molecular testing benefits grows across sectors, the demand for versatile RT-PCR kits is expected to rise. The challenges in the RT-PCR kit market are:
• High Costs and Limited Accessibility: The cost of RT-PCR kits and associated testing infrastructure remains a significant barrier, especially in low- and middle-income countries. High reagent costs, equipment expenses, and skilled labor requirements limit widespread adoption. This restricts market growth in resource-constrained settings and hampers efforts to achieve universal testing coverage, which is crucial during pandemics and for routine diagnostics.
• Regulatory and Quality Assurance Challenges: The market faces hurdles related to stringent regulatory approvals, quality control, and standardization of RT-PCR kits. Variability in regulatory requirements across regions can delay product launches and increase compliance costs. Ensuring consistent quality and performance of kits is vital for clinical reliability but remains challenging, impacting market confidence and adoption rates.
• Supply Chain Disruptions: The COVID-19 pandemic highlighted vulnerabilities in supply chains for raw materials and finished products. Disruptions in manufacturing, transportation, and distribution channels can lead to shortages and delays, affecting testing capacity and market stability. Ensuring a resilient supply chain is critical for maintaining steady market growth and meeting global testing demands. The RT-PCR kit market is driven by technological innovations, rising infectious disease prevalence, supportive government policies, and expanding applications. However, high costs, regulatory complexities, and supply chain issues pose significant challenges. These factors collectively influence market dynamics, requiring stakeholders to innovate and adapt to sustain growth. The overall impact suggests a promising yet complex landscape, with opportunities for growth tempered by operational and regulatory hurdles that must be strategically managed.
List of RT-PCR Kit Companies
Companies in the market compete on the basis of product quality offered. Major players in this market focus on expanding their manufacturing facilities, R&D investments, infrastructural development, and leverage integration opportunities across the value chain. With these strategies RT-PCR kit companies cater increasing demand, ensure competitive effectiveness, develop innovative products & technologies, reduce production costs, and expand their customer base. Some of the RT-PCR kit companies profiled in this report include-• Roche
• Thermo Fisher Scientific
• Bio-Rad
• Qiagen
• Takara Bio
• Agilent Technologies
• BioMérieux
• Standard BioTools
• Danaher
• Merck KGaA
RT-PCR Kit Market by Segment
The study includes a forecast for the global RT-PCR kit market by type, application, and region.RT-PCR Kit Market by Type [Value from 2019 to 2035]:
• Reverse Transcription Kits
• PCR Enhancement Kits
• Others
RT-PCR Kit Market by Application [Value from 2019 to 2035]:
• Disease Diagnosis
• Gene Expression Analysis
• Drug Discovery
• Others
RT-PCR Kit Market by Region [Value from 2019 to 2035]:
• North America
• Europe
• Asia Pacific
• The Rest of the World
Country Wise Outlook for the RT-PCR Kit Market
The RT-PCR kit market has experienced significant growth driven by advancements in diagnostic technologies, increased demand for rapid and accurate testing, and the ongoing need for effective disease management. The COVID-19 pandemic accelerated the adoption of RT-PCR testing worldwide, prompting innovations and expanding market reach. Countries are investing in research and development to improve test accuracy, reduce costs, and enhance accessibility. Regulatory approvals and collaborations between biotech firms and healthcare providers are also shaping the market landscape. As the global health landscape evolves, these developments reflect a focus on improving diagnostic capabilities and preparedness for future outbreaks.• United States: The US market for RT-PCR kits has seen rapid growth due to high healthcare expenditure and technological innovation. Major companies are developing faster, more accurate testing solutions, and government agencies are investing heavily in expanding testing capacity. The FDA has approved numerous RT-PCR kits, boosting market confidence. Additionally, the integration of automation and AI in testing processes has improved efficiency. The US also focuses on developing multiplex RT-PCR kits for simultaneous detection of multiple pathogens, enhancing diagnostic capabilities for infectious diseases beyond COVID-19.
• China: China has rapidly expanded its RT-PCR kit production capacity to meet domestic and export demands. The government has prioritized COVID-19 testing infrastructure, leading to increased adoption of advanced RT-PCR technologies. Local manufacturers are innovating with cost-effective, rapid testing kits to improve accessibility in rural and urban areas. China is also investing in research to develop multiplex and point-of-care RT-PCR solutions, aiming to enhance disease surveillance and control. Regulatory frameworks have been streamlined to facilitate faster approval processes, supporting market growth and technological advancements.
• Germany: Germanys RT-PCR kit market is characterized by high-quality standards and rigorous regulatory oversight. The country emphasizes precision medicine and personalized diagnostics, leading to the development of highly sensitive RT-PCR kits. German biotech firms are collaborating with academic institutions to innovate in multiplex testing and automation. The market benefits from strong healthcare infrastructure and government support for research initiatives. Additionally, Germany is focusing on expanding testing for emerging infectious diseases and integrating RT-PCR testing into broader public health strategies, ensuring preparedness and rapid response capabilities.
• India: India has experienced a surge in RT-PCR testing demand due to the COVID-19 pandemic, prompting local manufacturers to scale up production. The market is driven by government initiatives to increase testing accessibility in rural and underserved areas. Cost-effective, rapid RT-PCR kits are being developed to address affordability issues. The country is also investing in research to improve test accuracy and reduce turnaround times. Public-private partnerships are fostering innovation, and regulatory agencies are streamlining approval processes to facilitate market expansion. These efforts aim to strengthen Indias diagnostic infrastructure and disease management capabilities.
• Japan: Japans RT-PCR kit market is focused on high precision and reliability, with a strong emphasis on quality control. The country is investing in next-generation sequencing and multiplex RT-PCR technologies to enhance diagnostic accuracy. Japanese companies are developing portable and point-of-care testing solutions to improve accessibility in remote areas. The government supports research collaborations to innovate in infectious disease diagnostics and bioinformatics integration. Japan also emphasizes regulatory compliance and product safety, ensuring that RT-PCR kits meet stringent standards. These developments aim to bolster Japans capacity for early detection and effective management of infectious diseases.
Features of the Global RT-PCR Kit Market
Market Size Estimates: RT-PCR kit market size estimation in terms of value ($B). Trend and Forecast Analysis: Market trends (2019 to 2025) and forecast (2026 to 2035) by various segments and regions. Segmentation Analysis: RT-PCR kit market size by type, application, and region in terms of value ($B). Regional Analysis: RT-PCR kit market breakdown by North America, Europe, Asia Pacific, and Rest of the World. Growth Opportunities: Analysis of growth opportunities in different types, applications, and regions for the RT-PCR kit market. Strategic Analysis: This includes M&A, new product development, and competitive landscape of the RT-PCR kit market. Analysis of competitive intensity of the industry based on Porter’s Five Forces model.FAQ
Q1. What is the market size and growth forecast for RT-PCR kit market? Answer: The global RT-PCR kit market is expected to reach an estimated $6.5 billion by 2035 and grow with a CAGR of 5.9% from 2026 to 2035. Q2. What are the major drivers influencing the growth of the RT-PCR kit market? Answer: The major drivers for this market are the increasing demand for rapid disease detection, the rising prevalence of infectious diseases, and the growing adoption in clinical diagnostic laboratories. Q3. What are the major segments for RT-PCR kit market? Answer: The future of the RT-PCR kit market looks promising with opportunities in the disease diagnosis, gene expression analysis, and drug discovery markets. Q4. Who are the key RT-PCR kit market companies? Answer: Some of the key RT-PCR kit companies are as follows:• Roche
• Thermo Fisher Scientific
• Bio-Rad
• Qiagen
• Takara Bio
• Agilent Technologies
• BioMérieux
• Standard BioTools
• Danaher
• Merck KGaA Q5. Which RT-PCR kit market segment will be the largest in future? Answer: Lucintel forecasts that, within the type category, reverse transcription kit is expected to witness higher growth over the forecast period. Q6. In RT-PCR kit market, which region is expected to be the largest in next 5 years? Answer: In terms of region, APAC is expected to witness the highest growth over the forecast period. Q7. Do we receive customization in this report? Answer: Yes, Lucintel provides 10% customization without any additional cost.
Table of Contents
Methodology
- In-depth interviews of the major players in this market
- Detailed secondary research from competitors’ financial statements and published data
- Extensive searches of published works, market, and database information pertaining to industry news, company press releases, and customer intentions
- A compilation of the experiences, judgments, and insights of Lucintel’s professionals, who have analyzed and tracked this market over the years.
Buy Now
Choose a license that fits your team. Instant PDF delivery.
Prices exclude taxes. Instant delivery. Custom licensing available on request.
Key Questions
- • What are some of the most promising, high-growth opportunities for the RT-PCR kit market by type (reverse transcription kits, PCR enhancement kits, and others), application (disease diagnosis, gene expression analysis, drug discovery, and others), and region (North America, Europe, Asia Pacific, and the Rest of the World)?
- • Which segments will grow at a faster pace and why?
- • Which region will grow at a faster pace and why?
- • What are the key factors affecting market dynamics? What are the key challenges and business risks in this market?
- • What are the business risks and competitive threats in this market?
- • What are the emerging trends in this market and the reasons behind them?
- • What are some of the changing demands of customers in the market?
- • What are the new developments in the market? Which companies are leading these developments?
- • Who are the major players in this market? What strategic initiatives are key players pursuing for business growth?
- • What are some of the competing products in this market and how big of a threat do they pose for loss of market share by material or product substitution?
- • What M&A activity has occurred in the last 7 years and what has its impact been on the industry? For any questions related to RT-PCR Kit Market, RT-PCR Kit Market Size, RT-PCR Kit Market Growth, RT-PCR Kit Market Analysis, RT-PCR Kit Market Report, RT-PCR Kit Market Share, RT-PCR Kit Market Trends, RT-PCR Kit Market Forecast, RT-PCR Kit Companies, write Lucintel analyst at email: helpdesk@lucintel.com. We will be glad to get back to you soon.