Report Feature
Neonatal Light Therapy Device Market in Australia Trends and Forecast
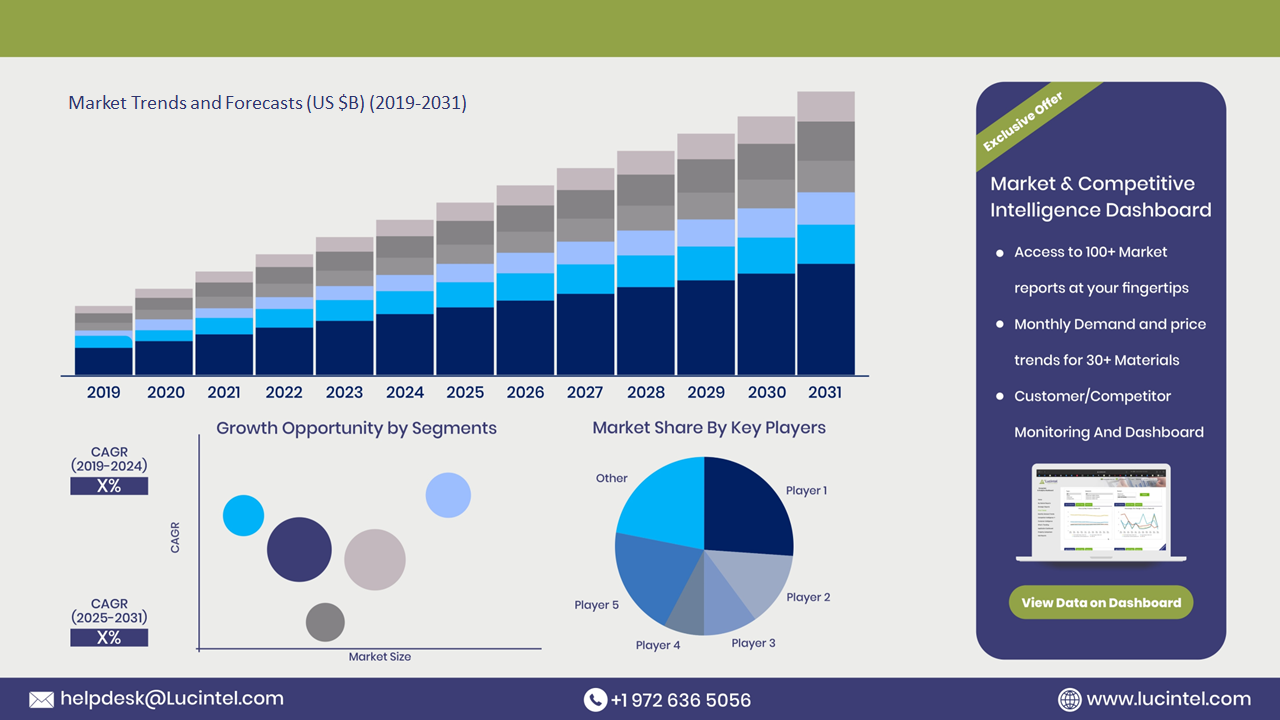
The future of the neonatal light therapy device market in Australia looks promising with opportunities in the hospital & clinic and birthing center markets. The global neonatal light therapy device market is expected to grow with a CAGR of 4.2% from 2025 to 2031. The neonatal light therapy device market in Australia is also forecasted to witness strong growth over the forecast period. The major drivers for this market are rising awareness among healthcare providers and increasing healthcare spending on neonatal care.• Lucintel forecasts that, within the type category, the phototherapy lamp is expected to witness higher growth over the forecast period.
• Within the application category, hospitals & clinics are expected to witness higher growth.
Emerging Trends in the Neonatal Light Therapy Device Market in Australia
The neonatal light therapy device market in Australia is experiencing rapid growth driven by advancements in medical technology, increasing awareness of neonatal jaundice treatment, and a rising birth rate. As healthcare providers seek more effective and safer solutions, innovative devices are emerging to meet these demands. The market is also influenced by government initiatives promoting neonatal health and the adoption of advanced medical equipment in hospitals. Additionally, technological integration, such as smart devices and portable solutions are gaining popularity, making treatment more accessible and efficient. These developments are transforming neonatal care, improving patient outcomes, and expanding market opportunities across Australia.• Technological Innovation: The market is witnessing the development of advanced light therapy devices featuring LED technology, which offers better energy efficiency, longer lifespan, and improved safety. These innovations enable more precise control of light intensity and wavelength, enhancing treatment efficacy. Portable and compact devices are also emerging, allowing for greater flexibility in neonatal care settings. The integration of smart technology with remote monitoring capabilities is further improving patient management. Overall, technological innovation is driving the adoption of more effective, safer, and user-friendly neonatal light therapy devices, reshaping treatment protocols.
• Increasing Awareness and Diagnosis: Growing awareness among healthcare professionals and parents about neonatal jaundice and its potential complications is fueling demand for effective treatment options. Early diagnosis through improved screening methods is leading to timely intervention, reducing the risk of long-term health issues. Educational campaigns and training programs are enhancing knowledge about neonatal light therapy benefits, encouraging its widespread use. As awareness increases, hospitals and clinics are more inclined to invest in advanced light therapy devices, which is expanding the market. This trend is crucial for early intervention and improving neonatal health outcomes across Australia.
• Regulatory Approvals and Standards: Stringent regulatory frameworks and standards set by Australian health authorities are influencing device development and market entry. Manufacturers are focusing on compliance with safety, efficacy, and quality standards to gain approval. This ensures that devices are safe for neonatal use and meet international benchmarks, boosting consumer confidence. Regulatory support also encourages innovation by providing clear pathways for new device approval. As standards evolve, companies are investing in research and development to meet these requirements, which ultimately enhances device reliability and market growth in Australia.
• Market Expansion and Hospital Adoption: The increasing number of hospitals and neonatal intensive care units (NICUs) across Australia is driving demand for neonatal light therapy devices. Hospitals are investing in advanced equipment to improve neonatal care quality and outcomes. The trend towards centralizing neonatal services and upgrading existing facilities is also contributing to market expansion. Additionally, private healthcare providers are adopting these devices to offer comprehensive neonatal services. This growth in hospital infrastructure and adoption is creating significant opportunities for device manufacturers, fostering a competitive and innovative market environment.
• Focus on Portable and Home-based Solutions: The demand for portable, easy-to-use neonatal light therapy devices is rising, driven by the need for flexible treatment options outside traditional hospital settings. These devices enable home-based care, reducing hospital stays and associated costs. They are particularly beneficial in rural and remote areas where access to specialized neonatal care is limited. The development of lightweight, battery-operated devices with user-friendly interfaces is making neonatal treatment more accessible. This trend is expanding market reach and improving neonatal health management by facilitating early and continuous treatment in diverse settings. These emerging trends are collectively reshaping the neonatal light therapy device market in Australia by fostering innovation, expanding access, and enhancing treatment efficacy. Technological advancements are making devices safer and more efficient, while increased awareness and regulatory support are driving market growth. The expansion of hospital infrastructure and the shift towards portable, home-based solutions are broadening the scope of neonatal care. As these trends continue to evolve, they will lead to improved neonatal health outcomes, greater market competitiveness, and a more patient-centric approach to neonatal treatment across Australia.
Recent Developments in the Neonatal Light Therapy Device Market in Australia
The neonatal light therapy device market in Australia is experiencing rapid growth driven by advancements in medical technology, increasing awareness of neonatal jaundice, and expanding healthcare infrastructure. As neonatal care becomes a priority, innovative devices are being adopted to improve treatment outcomes. The market is also influenced by government initiatives and rising healthcare expenditure, which facilitate the adoption of advanced therapies. Additionally, the growing prevalence of neonatal health issues necessitates effective and safe treatment options. These developments collectively are shaping a dynamic landscape that aims to enhance neonatal care quality across Australia.• Technological Innovations: New LED-based devices are improving treatment efficiency and safety. These devices offer better light spectrum control, energy efficiency, and portability, making neonatal jaundice management more effective. The integration of smart features allows real-time monitoring and data collection, enhancing patient care. Such innovations are reducing treatment duration and minimizing side effects, leading to better health outcomes for neonates. The adoption of these advanced devices is expected to increase, driven by healthcare providers seeking improved treatment options. Overall, technological progress is transforming neonatal light therapy, making it more accessible and effective.
• Rising Incidence of Neonatal Jaundice: The increasing prevalence of neonatal jaundice in Australia is fueling demand for effective light therapy devices. Factors such as premature births, genetic predispositions, and maternal health issues contribute to this rise. As awareness about early diagnosis and treatment grows, healthcare providers are investing more in advanced therapy options. This trend is prompting manufacturers to develop specialized devices tailored to neonatal needs. The heightened demand is also encouraging research and development efforts to improve device efficacy and safety. Consequently, the market is expanding rapidly to meet the growing healthcare needs of neonates with jaundice.
• Government Initiatives and Healthcare Policies: Australian government programs are promoting neonatal health through funding and policy support. These initiatives aim to improve neonatal care infrastructure and ensure access to advanced treatment devices across urban and rural areas. Public health campaigns are raising awareness about neonatal jaundice and the importance of early intervention. Additionally, regulatory frameworks are facilitating faster approval and adoption of innovative devices. These policies are creating a conducive environment for market growth by encouraging healthcare providers to adopt new technologies. Overall, government support is pivotal in expanding the reach and quality of neonatal light therapy services.
• Increasing Healthcare Expenditure: Rising healthcare spending in Australia is enabling hospitals and clinics to invest in modern neonatal treatment devices. This financial growth is driven by a focus on improving neonatal outcomes and reducing long-term health complications. Healthcare facilities are upgrading their equipment to incorporate the latest light therapy devices, which offer better safety and efficacy. The increased budget allocations also support training and capacity building for healthcare professionals. As a result, more neonates are receiving timely and effective treatment, which enhances overall neonatal health standards. This trend is expected to sustain market growth and innovation.
• Market Expansion and Strategic Collaborations: The neonatal light therapy device market is witnessing expansion through new entrants and strategic partnerships. Companies are collaborating with research institutions to develop next-generation devices. Market players are also expanding distribution networks to reach remote and underserved areas. These collaborations facilitate technological advancements and cost-effective solutions, making devices more accessible. The entry of international companies into the Australian market is further intensifying competition, leading to product innovation and price competitiveness. Overall, strategic alliances and market expansion are driving growth, ensuring wider availability of neonatal light therapy devices across Australia. These recent developments are significantly impacting the neonatal light therapy device market in Australia by fostering innovation, increasing accessibility, and improving neonatal health outcomes. Technological advancements and rising disease prevalence are driving demand, while government initiatives and healthcare expenditure support market expansion. Strategic collaborations are enhancing product offerings and distribution, ensuring broader reach. Collectively, these factors are creating a robust market environment that prioritizes neonatal health, encourages innovation, and ensures better treatment options for neonates across Australia.
Strategic Growth Opportunities in the Neonatal Light Therapy Device Market in Australia
The neonatal light therapy device market in Australia is experiencing rapid growth driven by increasing awareness of neonatal jaundice treatment, technological advancements, and rising healthcare investments. As neonatal care standards improve, the demand for efficient, safe, and portable light therapy devices is expanding across hospitals, clinics, and home care settings. Key applications such as hospital neonatal units, home care, and research institutions are fueling market expansion. Strategic growth opportunities are emerging through innovation, partnerships, and regional penetration, shaping the future landscape of neonatal care in Australia. These developments are poised to enhance patient outcomes, reduce treatment costs, and broaden access to advanced neonatal therapies.• Hospital Neonatal Units: Enhanced infrastructure and rising birth rates are driving demand for advanced light therapy devices in hospital neonatal units. These units require reliable, high-efficiency devices to treat neonatal jaundice effectively, ensuring better health outcomes. The focus on infection control and portability is prompting manufacturers to innovate, leading to safer, more user-friendly devices. This growth opportunity allows companies to expand their product portfolios and strengthen relationships with healthcare providers. As hospitals prioritize neonatal care quality, the adoption of cutting-edge light therapy solutions is expected to surge, significantly impacting market growth and patient care standards.
• Home Care Settings: Growing awareness of neonatal jaundice management at home is creating a significant market opportunity for portable, easy-to-use light therapy devices. Parents and caregivers seek cost-effective, safe solutions that enable early intervention and reduce hospital stays. The development of compact, battery-operated devices with remote monitoring capabilities is facilitating this shift. This trend not only broadens access to treatment but also alleviates healthcare system burdens. Companies investing in user-friendly, affordable home care devices are poised to capture a substantial share of this expanding segment, transforming neonatal jaundice management and improving patient comfort.
• Research and Clinical Applications: The increasing focus on neonatal research and clinical trials is driving demand for specialized light therapy devices with precise control features. These devices are essential for studying treatment efficacy, optimizing protocols, and developing new therapies. Investment in research infrastructure and collaborations with academic institutions are fueling this growth. The availability of advanced, customizable devices enhances research capabilities, leading to innovations in neonatal care. This opportunity supports the development of evidence-based practices and positions manufacturers as key players in neonatal health advancements, ultimately impacting clinical outcomes and treatment standards.
• Technological Innovation and Product Development: Continuous technological advancements, such as LED-based systems and smart device integration, are revolutionizing neonatal light therapy. These innovations improve energy efficiency, safety, and ease of use, while enabling real-time monitoring and data collection. Companies focusing on R&D to develop next-generation devices can differentiate themselves in a competitive market. The integration of IoT and AI technologies offers personalized treatment options and enhances clinical decision-making. This growth opportunity accelerates market expansion by providing more effective, safer, and user-centric solutions, ultimately elevating neonatal care quality across Australia.
• Regional Penetration and Market Expansion: Expanding distribution networks and regional outreach are critical for capturing untapped markets within Australia, especially in rural and remote areas. Developing affordable, portable devices suited for diverse healthcare settings can address regional disparities in neonatal care. Strategic partnerships with local healthcare providers and government initiatives can facilitate market penetration. This approach broadens access to neonatal light therapy, improves health outcomes in underserved populations, and stimulates overall market growth. Strengthening regional presence ensures comprehensive neonatal care coverage, fostering sustainable market development and innovation dissemination. These strategic growth opportunities are significantly impacting the neonatal light therapy device market in Australia by expanding access, enhancing device innovation, and improving neonatal care standards. Hospital and home care segments are experiencing increased adoption driven by technological advancements and rising awareness. Research and development efforts are fostering new treatment protocols, while regional expansion ensures broader healthcare reach. Collectively, these developments are propelling market growth, improving patient outcomes, and establishing Australia as a leader in neonatal healthcare innovation.
Neonatal Light Therapy Device Market in Australia Driver and Challenges
The neonatal light therapy device market in Australia is influenced by a variety of technological, economic, and regulatory factors. Advances in medical technology, increasing awareness of neonatal jaundice, and supportive healthcare policies are key drivers. Conversely, challenges such as high device costs, regulatory hurdles, and limited access in rural areas pose significant barriers. Understanding these drivers and challenges is essential for stakeholders aiming to optimize neonatal care and expand market reach. The interplay of these factors shapes the growth trajectory and innovation landscape within this specialized medical device sector. The factors responsible for driving the neonatal light therapy device market in Australia include:-• Technological Advancements: The continuous development of more efficient, portable, and user-friendly light therapy devices enhances treatment outcomes and patient comfort. Innovations such as LED-based devices offer energy efficiency and longer lifespan, making them more attractive to healthcare providers. These technological improvements reduce treatment time and improve neonatal safety, encouraging wider adoption. As technology evolves, the market is expected to see increased demand driven by improved device efficacy and ease of use, ultimately benefiting neonatal care standards across Australia.
• Rising Incidence of Neonatal Jaundice: The increasing prevalence of neonatal jaundice in Australia, driven by factors like premature births and genetic predispositions, is a major market driver. Early diagnosis and effective treatment are critical, and light therapy remains the primary intervention. Growing awareness among healthcare professionals and parents about the importance of timely treatment is fueling demand for advanced light therapy devices. This trend underscores the need for accessible, reliable devices, prompting manufacturers to innovate and expand their product offerings to meet the rising clinical demand.
• Government and Healthcare Policies: Supportive regulatory frameworks and government initiatives aimed at improving neonatal healthcare significantly impact market growth. Policies promoting early diagnosis and treatment, along with funding for neonatal intensive care units (NICUs), facilitate the adoption of light therapy devices. Additionally, Australia’s focus on reducing neonatal mortality rates encourages investments in advanced medical technologies. These policies create a conducive environment for market expansion, encouraging manufacturers to develop compliant, high-quality devices and ensuring broader access to neonatal light therapy across diverse healthcare settings.
• Economic Factors and Healthcare Spending: Australia’s robust healthcare infrastructure and increasing healthcare expenditure support the adoption of advanced neonatal treatment devices. Rising disposable incomes and government funding enable hospitals and clinics to invest in state-of-the-art light therapy equipment. Cost-effectiveness and long-term benefits of LED-based devices further influence purchasing decisions. As healthcare budgets grow, there is a greater scope for upgrading neonatal care facilities, which drives market growth. Economic stability thus plays a crucial role in fostering innovation and expanding access to neonatal light therapy solutions.
• Growing Awareness and Education: Increased awareness among healthcare professionals and parents about neonatal jaundice and its treatment options is a key driver. Educational campaigns and training programs improve understanding of the importance of early intervention, leading to higher device utilization. This heightened awareness encourages hospitals to invest in modern light therapy devices, ensuring better neonatal outcomes. As knowledge dissemination expands, demand for advanced, safe, and effective devices is expected to rise, further propelling market growth and encouraging innovation in device design and functionality. The challenges in the neonatal light therapy device market in Australia are:-
• High Device Costs: The cost of advanced light therapy devices remains a significant barrier, especially for smaller hospitals and clinics with limited budgets. While LED technology offers long-term savings, initial purchase prices can be prohibitive. This financial constraint limits widespread adoption, particularly in rural or underfunded healthcare settings. Additionally, ongoing maintenance and replacement costs can further strain budgets. Overcoming this challenge requires manufacturers to develop cost-effective solutions and for policymakers to consider subsidies or funding programs to improve access to essential neonatal care devices.
• Regulatory and Approval Processes: Stringent regulatory requirements and lengthy approval procedures in Australia can delay the introduction of new devices into the market. Compliance with safety and efficacy standards necessitates extensive testing and documentation, which can be time-consuming and costly. These hurdles may discourage innovation and slow down the availability of cutting-edge devices, impacting patient care. Streamlining regulatory pathways while maintaining safety standards is crucial to facilitate faster market entry and ensure that neonates benefit from the latest technological advancements.
• Limited Access in Rural Areas: Geographic and infrastructural challenges restrict access to neonatal light therapy devices in remote and rural regions of Australia. Limited healthcare facilities, a shortage of trained personnel, and logistical issues hinder the deployment of advanced neonatal care equipment. This disparity results in unequal treatment outcomes and hampers overall market growth. Addressing this challenge requires strategic initiatives such as mobile clinics, telemedicine integration, and government programs aimed at improving healthcare infrastructure and training in underserved areas. In summary, technological innovations, rising neonatal jaundice cases, supportive policies, economic growth, and increased awareness are driving the growth of Australia’s neonatal light therapy device market. However, high costs, regulatory complexities, and access issues pose significant challenges. These factors collectively influence market dynamics, with ongoing efforts needed to enhance device affordability, streamline approval processes, and improve rural healthcare access. Overall, overcoming these challenges while leveraging drivers will be essential for sustainable growth and improved neonatal outcomes in Australia.
List of Neonatal Light Therapy Device Market in Australia Companies
Companies in the market compete on the basis of product quality offered. Major players in this market focus on expanding their manufacturing facilities, R&D investments, infrastructural development, and leverage integration opportunities across the value chain. Through these strategies, neonatal light therapy device companies cater to increasing demand, ensure competitive effectiveness, develop innovative products & technologies, reduce production costs, and expand their customer base. Some of the neonatal light therapy device companies profiled in this report include:• Company 1
• Company 2
• Company 3
• Company 4
• Company 5
• Company 6
• Company 7
• Company 8
• Company 9
• Company 10
Neonatal Light Therapy Device Market in Australia by Segment
The study includes a forecast for the neonatal light therapy device market in Australia by type and application.Neonatal Light Therapy Device Market in Australia by Type [Value from 2019 to 2031]:
• Phototherapy Lamps
• Phototherapy Blankets
Neonatal Light Therapy Device Market in Australia by Application [Value from 2019 to 2031]:
• Hospitals & Clinics
• Birthing Centers
Features of the Neonatal Light Therapy Device Market in Australia
Market Size Estimates: Neonatal light therapy device in Australia market size estimation in terms of value ($B). Trend and Forecast Analysis: Market trends and forecasts by various segments. Segmentation Analysis: Neonatal light therapy device in Australia market size by type and application in terms of value ($B). Growth Opportunities: Analysis of growth opportunities in different type and application for the neonatal light therapy device in Australia. Strategic Analysis: This includes M&A, new product development, and competitive landscape of the neonatal light therapy device in Australia. Analysis of competitive intensity of the industry based on Porter’s Five Forces model.If you are looking to expand your business in this or adjacent markets, then contact us. We have done hundreds of strategic consulting projects in market entry, opportunity screening, due diligence, supply chain analysis, M & A, and more.
FAQ
Q1. What are the major drivers influencing the growth of the neonatal light therapy device market in Australia? Answer: The major drivers for this market are rising awareness among healthcare providers and increasing healthcare spending on neonatal care. Q2. What are the major segments for neonatal light therapy device market in Australia? Answer: The future of the neonatal light therapy device market in Australia looks promising with opportunities in the hospitals & clinics and birthing centers markets. Q3. Which neonatal light therapy device market segment in Australia will be the largest in future? Answer: Lucintel forecasts that, within the type category, the phototherapy lamp is expected to witness higher growth over the forecast period. Q4. Do we receive customization in this report? Answer: Yes, Lucintel provides 10% customization without any additional cost.Table of Contents
Methodology
- In-depth interviews of the major players in this market
- Detailed secondary research from competitors’ financial statements and published data
- Extensive searches of published works, market, and database information pertaining to industry news, company press releases, and customer intentions
- A compilation of the experiences, judgments, and insights of Lucintel’s professionals, who have analyzed and tracked this market over the years.
Buy Now
Choose a license that fits your team. Instant PDF delivery.
Prices exclude taxes. Instant delivery. Custom licensing available on request.
Key Questions
- • What are some of the most promising, high-growth opportunities for the neonatal light therapy device market in Australia by type (phototherapy lamps and phototherapy blankets) and application (hospitals & clinics and birthing centers)?
- • Which segments will grow at a faster pace and why?
- • What are the key factors affecting market dynamics? What are the key challenges and business risks in this market?
- • What are the business risks and competitive threats in this market?
- • What are the emerging trends in this market and the reasons behind them?
- • What are some of the changing demands of customers in the market?
- • What are the new developments in the market? Which companies are leading these developments?
- • Who are the major players in this market? What strategic initiatives are key players pursuing for business growth?
- • What are some of the competing products in this market and how big of a threat do they pose for loss of market share by material or product substitution?
- • What M&A activity has occurred in the last 5 years and what has its impact been on the industry? For any questions related to Neonatal Light Therapy Device Market in Australia, Neonatal Light Therapy Device Market Size, Neonatal Light Therapy Device Market in Australia Growth, Neonatal Light Therapy Device Market in Australia Analysis, Neonatal Light Therapy Device Market in Australia Report, Neonatal Light Therapy Device Market in Australia Share, Neonatal Light Therapy Device Market in Australia Trends, Neonatal Light Therapy Device Market in Australia Forecast, Neonatal Light Therapy Device Companies, write Lucintel analyst at email: helpdesk@lucintel.com. We will be glad to get back to you soon.