Report Feature
Endoscopic Reprocessing Medical Device Market in Australia Trends and Forecast
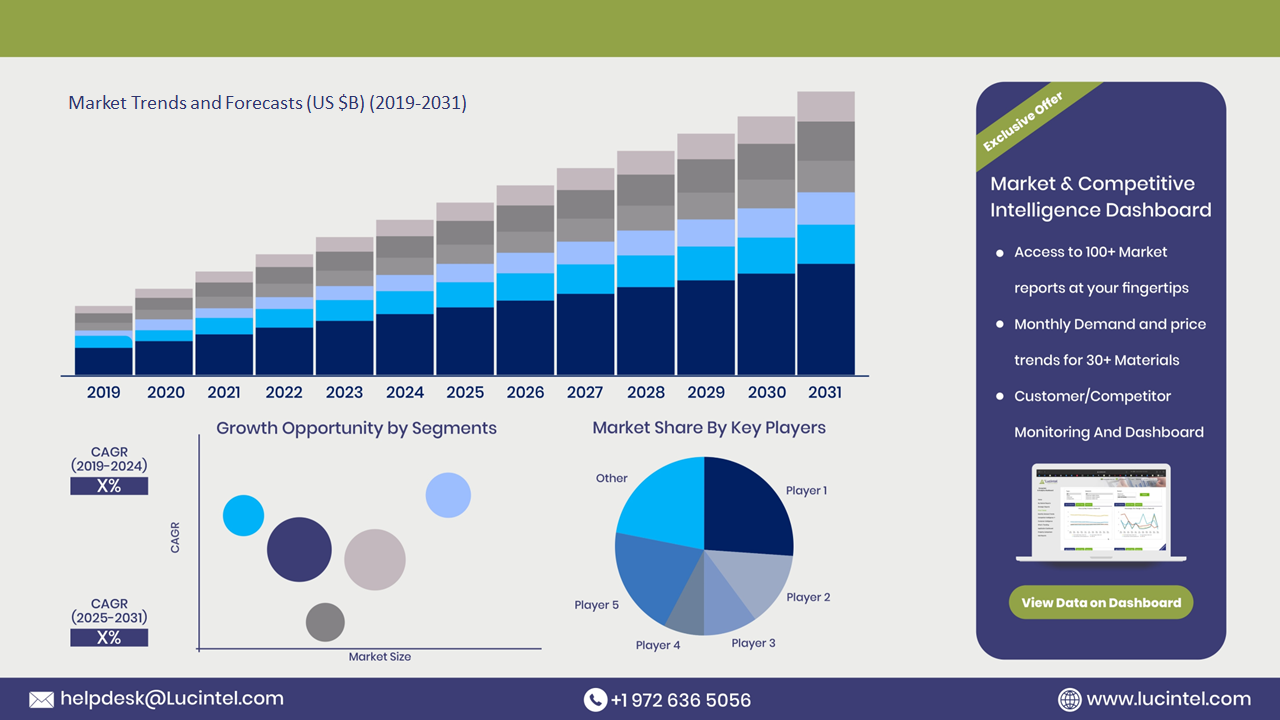
The future of the endoscopic reprocessing medical device market in Australia looks promising with opportunities in the hospital, ambulatory surgical center, and specialty clinic markets. The global endoscopic reprocessing medical device market is expected to grow with a CAGR of 7.8% from 2025 to 2031. The endoscopic reprocessing medical device market in Australia is also forecasted to witness strong growth over the forecast period. The major drivers for this market are the increasing demand for endoscopic procedures, the rising focus on infection control standards, and the growing integration of automation in reprocessing systems.• Lucintel forecasts that, within the type category, single chamber is expected to witness higher growth over the forecast period.
• Within the application category, hospital is expected to witness the highest growth.
Emerging Trends in the Endoscopic Reprocessing Medical Device Market in Australia
The endoscopic reprocessing medical device market in Australia is experiencing rapid growth driven by technological advancements, increasing healthcare standards, and a rising prevalence of gastrointestinal and respiratory conditions. As hospitals and clinics adopt more sophisticated sterilization techniques, the demand for efficient, safe, and eco-friendly reprocessing solutions is surging. Regulatory frameworks are also evolving to ensure patient safety, prompting manufacturers to innovate continuously. These developments are shaping a dynamic landscape that emphasizes quality, sustainability, and technological integration, ultimately improving patient outcomes and operational efficiency across healthcare facilities.• Technological Innovation: The integration of advanced sterilization technologies such as automated reprocessing systems and AI-driven monitoring tools is transforming the market. These innovations enhance efficiency, reduce human error, and ensure compliance with safety standards. As technology evolves, healthcare providers are increasingly adopting smarter, faster, and more reliable reprocessing solutions, which significantly improve patient safety and operational workflows.
• Regulatory and Safety Standards: Stricter government regulations and international safety standards are compelling healthcare providers to upgrade their reprocessing protocols. Compliance with these standards ensures patient safety and reduces infection risks. The market is witnessing increased investments in compliance-driven solutions, fostering innovation in sterilization methods and documentation processes, which ultimately enhance trust and safety in medical procedures.
• Rising Healthcare Expenditure: Australia’s growing healthcare expenditure, driven by an aging population and increased chronic disease prevalence, is fueling demand for endoscopic reprocessing devices. Hospitals and clinics are investing more in high-quality, durable, and efficient reprocessing equipment to meet rising patient care standards. This trend supports market expansion and encourages manufacturers to develop cost-effective, scalable solutions.
• Focus on Sustainability: Environmental concerns and sustainability initiatives are influencing market trends, prompting the adoption of eco-friendly reprocessing solutions. Manufacturers are developing energy-efficient sterilization devices and reducing chemical waste. Healthcare facilities are increasingly prioritizing sustainable practices, which not only reduce environmental impact but also align with global health and safety commitments.
• Digital Transformation and Data Management: The adoption of digital tools for tracking, documentation, and quality control in reprocessing is gaining momentum. Digital solutions improve traceability, compliance, and operational transparency. This trend enhances accountability, reduces errors, and streamlines workflows, making reprocessing more efficient and aligned with modern healthcare data management standards. These emerging trends are fundamentally reshaping the endoscopic reprocessing medical device market in Australia by fostering innovation, enhancing safety, and promoting sustainability. The integration of advanced technologies, stricter regulatory compliance, increased healthcare spending, eco-friendly practices, and digital transformation are collectively driving market growth. As these trends continue to evolve, they will improve patient outcomes, operational efficiency, and environmental responsibility, positioning Australia as a leader in safe and sustainable endoscopic reprocessing practices.
Recent Developments in the Endoscopic Reprocessing Medical Device Market in Australia
The endoscopic reprocessing medical device market in Australia is experiencing rapid growth driven by technological advancements, increasing healthcare demands, and stringent sterilization standards. As hospitals and clinics adopt innovative solutions to ensure patient safety, the market is expanding with new products and protocols. Regulatory updates and rising awareness about infection control are also fueling this growth. These developments are transforming healthcare practices, improving outcomes, and creating new opportunities for manufacturers and service providers in Australia’s medical sector.• Technological Innovations in Reprocessing Equipment: The introduction of automated and advanced reprocessing devices enhances efficiency and safety, reducing manual errors and contamination risks. These innovations improve turnaround times and compliance with strict sterilization standards, leading to increased adoption across healthcare facilities. The market benefits from increased demand for reliable, user-friendly devices that meet evolving regulatory requirements, ultimately elevating patient safety and operational efficiency.
• Regulatory and Policy Enhancements: Australian authorities have implemented stricter regulations and guidelines for endoscopic device sterilization, emphasizing patient safety and infection control. These policies compel healthcare providers to upgrade their reprocessing protocols and invest in compliant equipment. The regulatory landscape encourages innovation and standardization, fostering market growth as providers seek to meet compliance standards, reduce infection risks, and avoid penalties, thereby boosting demand for certified reprocessing devices.
• Rising Adoption of Single-Use Endoscopes: The shift towards single-use endoscopes reduces cross-contamination risks and simplifies reprocessing procedures. This trend is driven by concerns over infection transmission and the need for rapid turnover in busy healthcare settings. The market sees increased demand for disposable devices, which, while initially more costly, offer long-term savings in sterilization and infection management, influencing manufacturers to develop more cost-effective single-use options.
• Increasing Healthcare Infrastructure and Investment: Australia’s expanding healthcare infrastructure, including new hospitals and clinics, supports the adoption of advanced endoscopic reprocessing solutions. Government and private sector investments aim to improve healthcare quality and safety standards. This growth in infrastructure creates a larger customer base for reprocessing devices, encouraging innovation and competition, and ultimately leading to a broader availability of state-of-the-art sterilization technologies across the country.
• Growing Awareness and Training Initiatives: Enhanced training programs and awareness campaigns about infection control and proper reprocessing techniques are improving compliance among healthcare workers. These initiatives promote best practices, reduce errors, and increase demand for sophisticated reprocessing devices that are easier to operate and maintain. As knowledge spreads, the market benefits from increased adoption of high-quality, compliant reprocessing solutions, ensuring safer patient outcomes. The recent developments in the Australian endoscopic reprocessing medical device market are significantly impacting the industry by fostering innovation, improving safety standards, and expanding market opportunities. These trends are driving growth, encouraging regulatory compliance, and enhancing healthcare quality, ultimately benefiting patients and providers alike.
Strategic Growth Opportunities in the Endoscopic Reprocessing Medical Device Market in Australia
The endoscopic reprocessing medical device market in Australia is poised for significant growth driven by technological advancements, increasing healthcare investments, and rising awareness of infection control. The demand for efficient, safe, and compliant reprocessing solutions is expanding across hospitals, clinics, and ambulatory surgical centers. Strategic opportunities are emerging as stakeholders focus on improving patient outcomes, reducing procedural costs, and adhering to stringent regulatory standards. This evolving landscape offers numerous avenues for innovation, market penetration, and competitive differentiation.• Adoption of Advanced Reprocessing Technologies and Automation: The integration of automated reprocessing systems and cutting-edge technologies is transforming the Australian market. These innovations enhance sterilization efficiency, reduce manual errors, and ensure compliance with strict regulatory standards. Hospitals and clinics are increasingly investing in automated solutions to streamline workflows, improve turnaround times, and minimize infection risks. The shift towards digital and robotic reprocessing devices presents substantial growth opportunities for manufacturers aiming to meet evolving safety and efficiency demands.
• Rising Demand for Single-Use and Disposable Reprocessing Devices: The increasing preference for single-use and disposable reprocessing devices is driven by concerns over cross-contamination and infection control. These products eliminate the need for complex cleaning procedures, reduce turnaround times, and enhance patient safety. As healthcare facilities aim to comply with infection prevention protocols, the market for disposable reprocessing tools is expanding rapidly. Manufacturers focusing on innovative, cost-effective disposable solutions are well-positioned to capitalize on this trend.
• Implementation of Stringent Regulatory Standards and Compliance Measures: Australia’s strict regulatory environment for medical devices encourages the adoption of compliant reprocessing solutions. Growing emphasis on safety standards, quality assurance, and environmental regulations creates opportunities for companies to develop certified, high-quality products. Investment in R&D to meet evolving standards and obtain necessary approvals can provide a competitive edge. This regulatory landscape also fosters innovation in eco-friendly and sustainable reprocessing technologies.
• Expansion of Healthcare Infrastructure and Increasing Procedural Volumes: Australia’s expanding healthcare infrastructure, including new hospitals and outpatient centers, is driving demand for endoscopic reprocessing devices. The rising volume of endoscopic procedures, such as gastrointestinal and respiratory interventions, necessitates efficient reprocessing solutions. This growth is further supported by an aging population and increasing prevalence of chronic diseases, creating a sustained need for reliable, scalable reprocessing equipment across diverse healthcare settings.
• Growing Focus on Infection Control and Patient Safety Initiatives: Enhanced awareness of infection control protocols and patient safety is fueling demand for advanced reprocessing devices. Healthcare providers are prioritizing sterilization efficacy to prevent healthcare-associated infections (HAIs). Investments in training, quality management, and innovative sterilization technologies are expanding market opportunities. Companies that develop user-friendly, validated, and environmentally sustainable reprocessing solutions can gain a competitive advantage in this safety-conscious environment. In conclusion, these growth opportunities are set to significantly influence the Australian endoscopic reprocessing device market by fostering innovation, improving safety standards, and expanding healthcare infrastructure. Stakeholders who strategically invest in advanced technologies, compliance, and disposable solutions will be well-positioned to capitalize on the evolving demand. Overall, these trends will drive market expansion, enhance patient outcomes, and reinforce Australia’s position as a leader in endoscopic reprocessing solutions.
Endoscopic Reprocessing Medical Device Market in Australia Driver and Challenges
The endoscopic reprocessing medical device market in Australia is influenced by a variety of technological, economic, and regulatory factors. Advances in medical technology, increasing healthcare expenditure, and stringent safety standards are shaping the market’s growth trajectory. Additionally, rising awareness about infection control and the need for efficient sterilization processes are significant drivers. However, the market also faces challenges such as high equipment costs, regulatory complexities, and the need for specialized training. These factors collectively impact market dynamics, influencing adoption rates, innovation, and overall growth potential in Australia’s healthcare sector. The factors responsible for driving the endoscopic reprocessing medical device market in Australia include:-• Technological Innovation: The continuous development of advanced reprocessing devices, such as automated systems and improved sterilization techniques, enhances efficiency and safety. These innovations reduce manual errors, improve turnaround times, and ensure compliance with strict health standards. As hospitals and clinics adopt these technologies, market growth accelerates. The push for minimally invasive procedures also increases demand for reliable reprocessing solutions, making technological advancements a key growth driver.
• Increasing Healthcare Expenditure: Australia’s rising healthcare spending supports the adoption of sophisticated medical devices, including endoscopic reprocessing equipment. Governments and the private sector are investing heavily in healthcare infrastructure, which includes upgrading sterilization facilities. This financial commitment ensures better infection control, patient safety, and compliance with international standards, thereby fueling market expansion. The growing elderly population and prevalence of chronic diseases further amplify the need for advanced endoscopic procedures and reprocessing solutions.
• Stringent Regulatory Standards: Australian health authorities enforce strict regulations regarding infection control and device sterilization. Compliance with standards such as those set by the Therapeutic Goods Administration (TGA) and international guidelines compels healthcare providers to adopt high-quality reprocessing devices. These regulations drive innovation and ensure market players develop safer, more effective solutions. While regulatory compliance can be challenging, it ultimately promotes market integrity and patient safety, fostering trust and growth.
• Rising Incidence of Gastrointestinal and Respiratory Diseases: The increasing prevalence of conditions requiring endoscopic procedures, such as gastrointestinal disorders and respiratory issues, boosts demand for endoscopic devices. As these procedures become more common, the need for effective reprocessing solutions grows to prevent cross-contamination and infections. This trend supports market expansion, especially in outpatient and hospital settings, where infection control is paramount.
• Growing Adoption of Minimally Invasive Procedures: The shift towards minimally invasive surgeries, driven by benefits like reduced recovery time and lower complication rates, increases the use of endoscopic devices. Consequently, the demand for reliable reprocessing equipment rises to ensure safety and compliance. This trend encourages manufacturers to innovate and expand their product portfolios, further propelling market growth in Australia. The challenges in the endoscopic reprocessing medical device market in Australia are:
• High Equipment and Maintenance Costs: The initial investment for advanced reprocessing systems is substantial, which can be a barrier for smaller healthcare facilities. Ongoing maintenance, calibration, and consumables add to operational expenses, potentially limiting adoption. These costs can strain budgets, especially in public hospitals with limited funding, thereby slowing market growth and creating disparities in access to cutting-edge reprocessing technology.
• Regulatory Complexities: Navigating Australia’s regulatory landscape, including compliance with TGA standards and international guidelines, can be complex and time-consuming. Stringent approval processes and frequent updates to regulations require continuous adaptation by manufacturers and healthcare providers. This complexity can delay product launches, increase costs, and hinder innovation, ultimately impacting market growth and the availability of new solutions.
• Need for Specialized Training: Proper operation and maintenance of advanced reprocessing devices require trained personnel. The shortage of skilled staff and the need for ongoing training pose challenges for healthcare facilities. Inadequate training can lead to improper sterilization, risking infections and regulatory non-compliance. Addressing this challenge involves investing in education and certification programs, which can be resource-intensive but are essential for ensuring safety and efficacy. In summary, technological advancements, increased healthcare spending, and regulatory compliance are key drivers propelling the Australian endoscopic reprocessing market. Conversely, high costs, regulatory hurdles, and training requirements pose significant challenges. Together, these factors shape a dynamic landscape that influences market growth, innovation, and safety standards. The overall impact is a market poised for growth, driven by technological progress and increasing procedural demand, but tempered by economic and operational hurdles that must be addressed for sustained expansion.
List of Endoscopic Reprocessing Medical Device Market in Australia Companies
Companies in the market compete on the basis of product quality offered. Major players in this market focus on expanding their manufacturing facilities, R&D investments, infrastructural development, and leverage integration opportunities across the value chain. Through these strategies, endoscopic reprocessing medical device companies cater to increasing demand, ensure competitive effectiveness, develop innovative products & technologies, reduce production costs, and expand their customer base. Some of the endoscopic reprocessing medical device companies profiled in this report include:• Company 1
• Company 2
• Company 3
• Company 4
• Company 5
• Company 6
• Company 7
• Company 8
• Company 9
• Company 10
Endoscopic Reprocessing Medical Device Market in Australia by Segment
The study includes a forecast for the endoscopic reprocessing medical device market in Australia by type and application.Endoscopic Reprocessing Medical Device Market in Australia by Type [Value from 2019 to 2031]:
• Single Chamber
• Multi Chamber
Endoscopic Reprocessing Medical Device Market in Australia by Application [Value from 2019 to 2031]:
• Hospitals
• Ambulatory Surgical Center
• Specialty Clinics
Features of the Endoscopic Reprocessing Medical Device Market in Australia
Market Size Estimates: Endoscopic reprocessing medical device in Australia market size estimation in terms of value ($B). Trend and Forecast Analysis: Market trends and forecasts by various segments. Segmentation Analysis: Endoscopic reprocessing medical device in Australia market size by type and application in terms of value ($B). Growth Opportunities: Analysis of growth opportunities in different type and application for the endoscopic reprocessing medical device in Australia. Strategic Analysis: This includes M&A, new product development, and competitive landscape of the endoscopic reprocessing medical device in Australia. Analysis of competitive intensity of the industry based on Porter’s Five Forces model.If you are looking to expand your business in this or adjacent markets, then contact us. We have done hundreds of strategic consulting projects in market entry, opportunity screening, due diligence, supply chain analysis, M & A, and more.
FAQ
Q1. What are the major drivers influencing the growth of the endoscopic reprocessing medical device market in Australia? Answer: The major drivers for this market are the increasing demand for endoscopic procedures, the rising focus on infection control standards, and the growing integration of automation in reprocessing systems. Q2. What are the major segments for endoscopic reprocessing medical device market in Australia? Answer: The future of the endoscopic reprocessing medical device market in Australia looks promising with opportunities in the hospital, ambulatory surgical center, and specialty clinic markets. Q3. Which endoscopic reprocessing medical device market segment in Australia will be the largest in future? Answer: Lucintel forecasts that, within the type category, single chamber is expected to witness higher growth over the forecast period. Q4. Do we receive customization in this report? Answer: Yes, Lucintel provides 10% customization without any additional cost.Table of Contents
Methodology
- In-depth interviews of the major players in this market
- Detailed secondary research from competitors’ financial statements and published data
- Extensive searches of published works, market, and database information pertaining to industry news, company press releases, and customer intentions
- A compilation of the experiences, judgments, and insights of Lucintel’s professionals, who have analyzed and tracked this market over the years.
Buy Now
Choose a license that fits your team. Instant PDF delivery.
Prices exclude taxes. Instant delivery. Custom licensing available on request.
Key Questions
- • What are some of the most promising, high-growth opportunities for the endoscopic reprocessing medical device market in Australia by type (single chamber and multi chamber) and application (hospitals, ambulatory surgical center, and specialty clinics)?
- • Which segments will grow at a faster pace and why?
- • What are the key factors affecting market dynamics? What are the key challenges and business risks in this market?
- • What are the business risks and competitive threats in this market?
- • What are the emerging trends in this market and the reasons behind them?
- • What are some of the changing demands of customers in the market?
- • What are the new developments in the market? Which companies are leading these developments?
- • Who are the major players in this market? What strategic initiatives are key players pursuing for business growth?
- • What are some of the competing products in this market and how big of a threat do they pose for loss of market share by material or product substitution?
- • What M&A activity has occurred in the last 5 years and what has its impact been on the industry? For any questions related to Endoscopic Reprocessing Medical Device Market in Australia, Endoscopic Reprocessing Medical Device Market in Australia Size, Endoscopic Reprocessing Medical Device Market in Australia Growth, Endoscopic Reprocessing Medical Device Market in Australia Analysis, Endoscopic Reprocessing Medical Device Market in Australia Report, Endoscopic Reprocessing Medical Device Market in Australia Share, Endoscopic Reprocessing Medical Device Market in Australia Trends, Endoscopic Reprocessing Medical Device Market in Australia Forecast, Endoscopic Reprocessing Medical Device Companies, write Lucintel analyst at email: helpdesk@lucintel.com. We will be glad to get back to you soon.