Report Feature
Acefylline Piperazine Market in United States Trends and Forecast
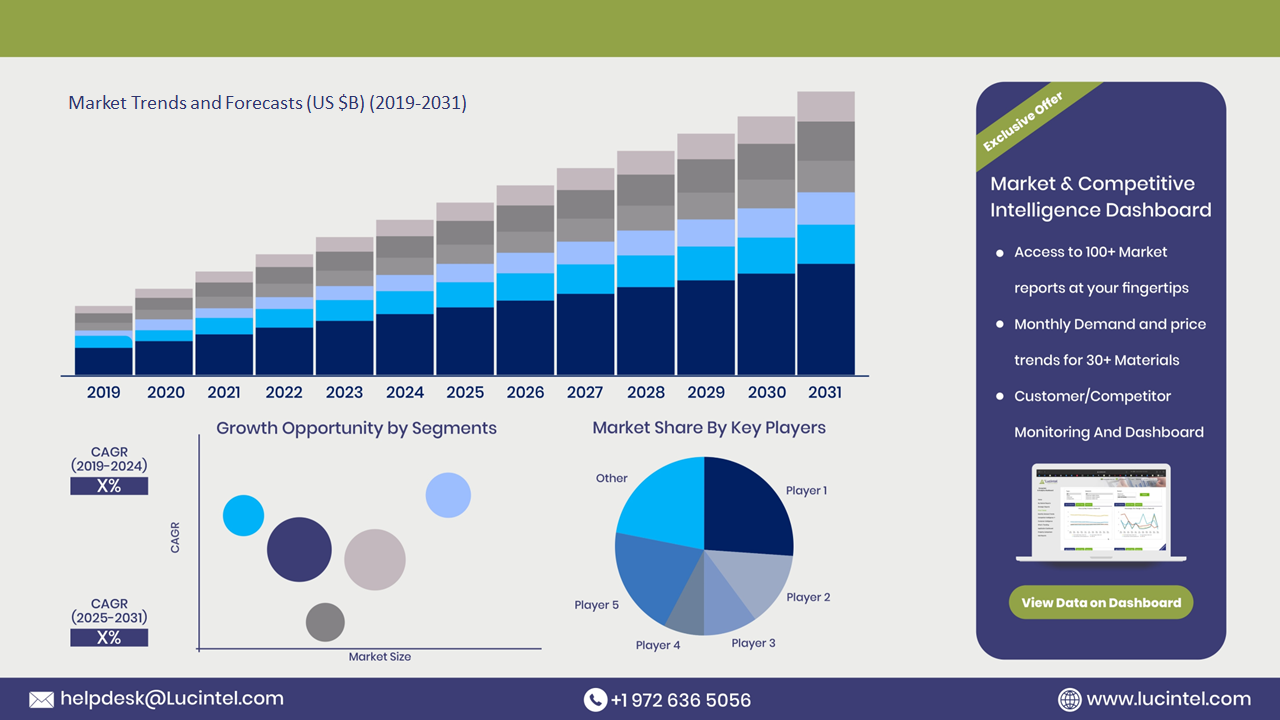
The future of the acefylline piperazine market in United States looks promising with opportunities in the COPD, asthma, and infant apnea markets. The global acefylline piperazine market is expected to grow with a CAGR of 2.3% from 2025 to 2031. The acefylline piperazine market in United States is also forecasted to witness strong growth over the forecast period. The major drivers for this market are the rising prevalence of respiratory & cardiovascular diseases, the growing adoption of combination therapies, and the increasing investment in pharmaceutical r&d.• Lucintel forecasts that, within the type category, purity 99% is expected to witness the highest growth over the forecast period.
• Within the application category, asthma is expected to witness the highest growth due to the rising awareness and early diagnosis.
Emerging Trends in the Acefylline Piperazine Market in United States
The acefylline piperazine market in the United States is experiencing rapid evolution driven by technological advancements, changing consumer preferences, and regulatory shifts. As healthcare providers seek more effective treatments, innovative formulations and delivery methods are emerging. Market players are also focusing on personalized medicine and digital health integration. These developments are shaping a dynamic landscape that demands agility and strategic foresight from stakeholders. Understanding these trends is crucial for capitalizing on new opportunities and maintaining a competitive advantage in this evolving sector.• Increasing Adoption of Personalized Medicine: Tailoring treatments based on genetic and biomarker data enhances efficacy and reduces side effects. This trend is driven by advancements in genomics and diagnostics, enabling more targeted therapies. It improves patient outcomes and fosters innovation in drug development, making treatments more precise and effective.
• Growth of Digital Health Integration: Incorporating telemedicine, mobile apps, and wearable devices into treatment plans enhances patient engagement and monitoring. Digital tools facilitate real-time data collection and remote consultations, improving treatment adherence and outcomes. This trend is transforming healthcare delivery and expanding access to care.
• Regulatory Landscape Evolution: Stricter regulations and approval processes are shaping market entry strategies. Agencies are emphasizing safety, efficacy, and transparency, which increases development timelines and costs. Companies must adapt to these changes by investing in compliance and robust clinical trials to succeed.
• Focus on Sustainable and Eco-friendly Formulations: Environmental concerns are prompting the development of greener manufacturing processes and biodegradable packaging. This shift aligns with consumer preferences for sustainable products and regulatory pressures. It enhances brand reputation and reduces environmental impact, fostering long-term market sustainability.
• Rising Demand for Combination Therapies: Combining Acefylline Piperazine with other agents offers enhanced therapeutic benefits and addresses complex conditions. This approach improves treatment efficacy and reduces medication burden. It encourages innovation in formulation and clinical research, expanding market opportunities. These emerging trends are fundamentally reshaping the acefylline piperazine market in the United States by fostering innovation, improving patient outcomes, and emphasizing sustainability. Digital integration and personalized medicine are enhancing treatment precision and accessibility. Regulatory adaptations ensure safety and efficacy, while sustainable practices align with societal values. Collectively, these developments are creating a more dynamic, patient-centric, and environmentally conscious market landscape, offering new opportunities for growth and competitive advantage.
Recent Developments in the Acefylline Piperazine Market in United States
The acefylline piperazine market in the United States is experiencing notable growth driven by advancements in pharmaceutical research and increasing demand for respiratory and central nervous system medications. Innovations in drug formulations and rising healthcare awareness are further fueling market expansion. Regulatory support and strategic collaborations among pharmaceutical companies are also contributing to this upward trajectory. As the market evolves, key players are focusing on developing targeted therapies to meet the growing needs of patients, thereby creating new opportunities for growth and innovation.• Growing Demand for Respiratory Medications: The increasing prevalence of respiratory conditions such as asthma and COPD in the U.S. has driven demand for Acefylline Piperazine-based therapies, which are effective in managing symptoms. This trend is supported by rising healthcare awareness and improved diagnostic techniques, leading to higher prescription rates. The market benefits from ongoing research to enhance drug efficacy and safety profiles, attracting investments from pharmaceutical companies. Consequently, this growth opportunity is expanding the market’s reach and encouraging innovation in respiratory treatments.
• Advances in Drug Delivery Systems: Innovations in drug delivery, including sustained-release formulations and inhalation therapies, are transforming how Acefylline Piperazine is administered. These advancements improve patient compliance, reduce side effects, and enhance therapeutic outcomes. The development of novel delivery mechanisms is attracting pharmaceutical companies to invest in research and development, fostering competitive advantages. As a result, the market is witnessing increased adoption of advanced delivery systems, which are expected to boost sales and expand the therapeutic applications of Acefylline Piperazine.
• Rising Focus on Central Nervous System Disorders: The increasing incidence of CNS disorders such as ADHD, narcolepsy, and depression in the U.S. has created a significant demand for effective medications. Acefylline Piperazine’s stimulant properties make it a promising candidate for managing these conditions. Pharmaceutical companies are investing in clinical trials to explore new indications, which could lead to expanded market applications. This focus on CNS disorders is driving innovation, regulatory approvals, and market growth, positioning Acefylline Piperazine as a vital component in neuropharmacology.
• Strategic Collaborations and Partnerships: Major pharmaceutical firms are forming alliances to accelerate the development and commercialization of Acefylline Piperazine products. These collaborations facilitate knowledge sharing, resource pooling, and faster regulatory approvals. Strategic partnerships also enable companies to expand their product portfolios and access new markets. Such collaborations are crucial for overcoming R&D challenges and driving innovation, ultimately strengthening the market position of Acefylline Piperazine and fostering sustainable growth within the U.S. healthcare landscape.
• Regulatory Advancements and Market Approvals: Recent regulatory approvals and supportive policies have streamlined the pathway for Acefylline Piperazine products to reach the market. Enhanced regulatory frameworks ensure safety and efficacy, boosting confidence among healthcare providers and patients. These developments encourage pharmaceutical companies to invest in research and expedite product launches. As a result, the market is witnessing increased product availability, which is expected to stimulate further growth, innovation, and competition in the U.S. acefylline piperazine market. The overall impact of these developments is a robust expansion of the acefylline piperazine market in the United States, driven by innovation, strategic collaborations, and regulatory support. These factors are enhancing product pipelines, improving patient outcomes, and increasing market competitiveness, ultimately fostering sustained growth and positioning Acefylline Piperazine as a key player in the pharmaceutical landscape.
Strategic Growth Opportunities in the Acefylline Piperazine Market in United States
The acefylline piperazine market in the United States is poised for significant expansion driven by increasing demand for respiratory and central nervous system medications. Advances in pharmaceutical research, rising prevalence of respiratory disorders, and a focus on innovative drug formulations are key factors fueling growth. Additionally, strategic collaborations and regulatory approvals are creating new opportunities for market players. This environment offers substantial potential for companies to capitalize on unmet medical needs and expand their product portfolios, ultimately enhancing healthcare outcomes and market competitiveness.• Growing Demand for Respiratory Medications: The rising incidence of respiratory conditions such as asthma and COPD in the U.S. is boosting the need for effective bronchodilators and respiratory stimulants like acefylline piperazine. Increased awareness and diagnosis rates are encouraging pharmaceutical companies to develop and market targeted therapies, expanding the overall market size. The focus on improving patient quality of life and managing chronic respiratory diseases further accelerates this growth opportunity.
• Increasing Prevalence of Neurological Disorders: The rising cases of neurological conditions such as Parkinson’s disease and depression are creating demand for central nervous system (CNS) stimulants and adjunct therapies. Acefylline piperazine’s potential neurostimulant properties make it a promising candidate for new drug formulations. Pharmaceutical companies are investing in research to explore their efficacy, which could lead to novel treatments and market expansion, especially as the aging population drives increased healthcare needs.
• Innovation in Drug Delivery Systems: Advances in drug delivery technologies, including controlled-release formulations and inhalation devices, are enhancing the effectiveness and patient compliance of acefylline piperazine-based therapies. These innovations enable targeted delivery, reduce side effects, and improve therapeutic outcomes. Companies investing in such technologies can differentiate their products, capture larger market shares, and meet evolving patient preferences, thereby creating a significant growth avenue within the U.S. pharmaceutical landscape.
• Strategic Collaborations and Licensing Agreements: Partnerships between pharmaceutical firms, biotech companies, and research institutions are facilitating the development of new acefylline piperazine formulations. Licensing deals and joint ventures accelerate product development timelines and expand market reach. These collaborations also enable access to advanced research, regulatory expertise, and distribution channels, which are crucial for capturing market share and driving revenue growth in a competitive environment.
• Regulatory Approvals and Market Entry Strategies: Securing FDA approval for new acefylline piperazine formulations is critical for market penetration. Companies focusing on compliance, clinical trial success, and strategic market entry plans can gain a competitive edge. Streamlined approval processes and favorable regulatory policies further support rapid commercialization, opening up opportunities for early market entry, increased sales, and establishing a strong foothold in the U.S. pharmaceutical sector. The overall impact of these growth opportunities is expected to significantly enhance the market landscape, fostering innovation, increasing accessibility to advanced therapies, and addressing unmet medical needs. As companies leverage these opportunities, the market will experience robust expansion, improved healthcare outcomes, and a stronger competitive environment, ultimately benefiting patients and stakeholders alike.
Acefylline Piperazine Market in United States Driver and Challenges
The acefylline piperazine market in the United States is influenced by a variety of technological, economic, and regulatory factors. Advances in pharmaceutical research and development are driving innovation, while economic conditions such as healthcare spending impact market growth. Regulatory frameworks shape product approval processes and market entry strategies, and technological progress enhances manufacturing efficiency and drug delivery systems. These interconnected factors create a dynamic environment that influences market expansion, competition, and innovation, ultimately affecting the availability and accessibility of Acefylline Piperazine-based medications in the United States. The factors responsible for driving the acefylline piperazine market in United States include:-• Technological Advancements: The continuous development of new drug formulations and delivery methods enhances the efficacy and safety of Acefylline Piperazine. Innovations such as targeted delivery systems and improved synthesis techniques reduce side effects and improve patient compliance. These technological improvements enable pharmaceutical companies to develop more effective products, expand their market share, and meet the evolving needs of healthcare providers and patients. Additionally, advancements in analytical and manufacturing technologies ensure higher quality standards, fostering trust and regulatory approval. Overall, technological progress accelerates product development cycles and broadens application scopes, fueling market growth.
• Increasing Healthcare Expenditure: The rising healthcare expenditure in the United States supports the development, approval, and distribution of new medications, including Acefylline Piperazine. Higher investment in healthcare infrastructure and research allows pharmaceutical companies to conduct extensive clinical trials and bring innovative products to market. Moreover, increased spending on chronic disease management and symptomatic relief drives demand for medications containing Acefylline Piperazine. This economic trend also facilitates better access to healthcare services, enabling more patients to benefit from these medications. Consequently, the expanding healthcare budget directly correlates with increased market opportunities and revenue potential.
• Regulatory Approvals and Policies: Stringent regulatory frameworks by agencies such as the FDA influence the market landscape. Clear guidelines for drug safety, efficacy, and manufacturing standards ensure high-quality products but can also pose barriers to entry for new players. Regulatory approvals are crucial for market access, and delays or rejections can hinder growth. Conversely, supportive policies and fast-track approval processes for innovative drugs can accelerate market penetration. Compliance with evolving regulations requires significant investment but ultimately ensures consumer safety and product credibility, shaping the competitive environment and influencing market dynamics.
• Growing Prevalence of Respiratory and Neurological Disorders: The increasing incidence of respiratory conditions and neurological disorders in the United States boosts demand for medications like Acefylline Piperazine, which are used to manage symptoms. Factors such as aging populations, environmental pollution, and lifestyle changes contribute to this trend. As healthcare providers seek effective symptomatic treatments, pharmaceutical companies focus on developing and marketing drugs that address these needs. This growing prevalence not only expands the target patient base but also encourages ongoing research and innovation, ensuring sustained market growth and diversification of product portfolios.
• Strategic Collaborations and Mergers: Partnerships between pharmaceutical companies, research institutions, and biotech firms facilitate the development and commercialization of Acefylline Piperazine products. Collaborations enable resource sharing, access to new technologies, and accelerated clinical trials, reducing time-to-market. Mergers and acquisitions also consolidate market power, enhance R&D capabilities, and expand distribution networks. These strategic moves help companies stay competitive amid evolving market conditions and regulatory landscapes. Overall, such collaborations and consolidations drive innovation, improve product pipelines, and support market expansion. The challenges in the acefylline piperazine market in United States are:
• Regulatory Hurdles: Navigating complex regulatory requirements can delay product approval and increase costs. The FDA’s stringent safety and efficacy standards demand extensive clinical data, which can be time-consuming and expensive to generate. Any delays or rejections impact market entry and revenue streams. Additionally, evolving regulations require continuous compliance efforts, posing ongoing challenges for manufacturers. Smaller companies may find these hurdles particularly daunting, limiting innovation and market diversity. Ensuring regulatory adherence while maintaining competitive pricing and timely product launches remains a significant challenge for stakeholders.
• Patent Expirations and Generic Competition: The expiration of patents for key Acefylline Piperazine formulations opens the market to generic competitors, leading to price erosion and reduced profit margins. Companies must innovate continuously to maintain market share, which involves high R&D costs. The influx of generics also intensifies price competition, impacting revenue streams. This challenge necessitates strategic planning around product lifecycle management, brand differentiation, and pipeline development to sustain profitability in a highly competitive environment.
• Supply Chain Disruptions: Disruptions in raw material supply, manufacturing processes, or distribution channels can significantly impact market stability. Factors such as geopolitical tensions, pandemics, or logistical issues can cause delays and shortages. These disruptions increase costs and reduce product availability, affecting patient access and company revenues. Ensuring a resilient supply chain requires substantial investment in diversified sourcing and inventory management. Overcoming these challenges is critical to maintaining a consistent product supply and market confidence amid unpredictable global conditions. In summary, technological innovations, economic growth, regulatory frameworks, disease prevalence, and strategic collaborations are key drivers propelling the acefylline piperazine market in the United States. However, regulatory hurdles, patent expirations, and supply chain issues pose significant challenges. Together, these factors shape a competitive, evolving landscape that demands continuous innovation and strategic agility. The overall impact is a dynamic market with substantial growth potential, driven by advancements and constrained by regulatory and operational hurdles, requiring stakeholders to adapt proactively for sustained success.
List of Acefylline Piperazine Market in United States Companies
Companies in the market compete on the basis of product quality offered. Major players in this market focus on expanding their manufacturing facilities, R&D investments, infrastructural development, and leverage integration opportunities across the value chain. Through these strategies, acefylline piperazine companies cater to increasing demand, ensure competitive effectiveness, develop innovative products & technologies, reduce production costs, and expand their customer base. Some of the acefylline piperazine companies profiled in this report include:• Company 1
• Company 2
• Company 3
• Company 4
• Company 5
• Company 6
• Company 7
Acefylline Piperazine Market in United States by Segment
The study includes a forecast for the acefylline piperazine market in United States by type and application.Acefylline Piperazine Market in United States by Type [Value from 2019 to 2031]:
• Purity 97%
• Purity 98%
• Purity 99%
• Others
Acefylline Piperazine Market in United States by Application [Value from 2019 to 2031]:
• COPD
• Asthma
• Infant Apnea
Features of the Acefylline Piperazine Market in United States
Market Size Estimates: Acefylline piperazine in United States market size estimation in terms of value ($B). Trend and Forecast Analysis: Market trends and forecasts by various segments. Segmentation Analysis: Acefylline piperazine in United States market size by type and application in terms of value ($B). Growth Opportunities: Analysis of growth opportunities in different type and application for the acefylline piperazine in United States. Strategic Analysis: This includes M&A, new product development, and competitive landscape of the acefylline piperazine in United States. Analysis of competitive intensity of the industry based on Porter’s Five Forces model.If you are looking to expand your business in this or adjacent markets, then contact us. We have done hundreds of strategic consulting projects in market entry, opportunity screening, due diligence, supply chain analysis, M & A, and more.
FAQ
Q1. What are the major drivers influencing the growth of the acefylline piperazine market in United States? Answer: The major drivers for this market are the rising prevalence of respiratory & cardiovascular diseases, the growing adoption of combination therapies, and the increasing investment in pharmaceutical r&d. Q2. What are the major segments for acefylline piperazine market in United States? Answer: The future of the acefylline piperazine market in United States looks promising with opportunities in the COPD, asthma, and infant apnea markets. Q3. Which acefylline piperazine market segment in United States will be the largest in future? Answer: Lucintel forecasts that, within the type category, purity 99% is expected to witness the highest growth over the forecast period. Q4. Do we receive customization in this report? Answer: Yes, Lucintel provides 10% customization without any additional cost.Table of Contents
Methodology
- In-depth interviews of the major players in this market
- Detailed secondary research from competitors’ financial statements and published data
- Extensive searches of published works, market, and database information pertaining to industry news, company press releases, and customer intentions
- A compilation of the experiences, judgments, and insights of Lucintel’s professionals, who have analyzed and tracked this market over the years.
Buy Now
Choose a license that fits your team. Instant PDF delivery.
Prices exclude taxes. Instant delivery. Custom licensing available on request.
Key Questions
- • What are some of the most promising, high-growth opportunities for the acefylline piperazine market in United States by type (purity 97%, purity 98%, purity 99%, and others) and application (COPD, asthma, and infant apnea)?
- • Which segments will grow at a faster pace and why?
- • What are the key factors affecting market dynamics? What are the key challenges and business risks in this market?
- • What are the business risks and competitive threats in this market?
- • What are the emerging trends in this market and the reasons behind them?
- • What are some of the changing demands of customers in the market?
- • What are the new developments in the market? Which companies are leading these developments?
- • Who are the major players in this market? What strategic initiatives are key players pursuing for business growth?
- • What are some of the competing products in this market and how big of a threat do they pose for loss of market share by material or product substitution?
- • What M&A activity has occurred in the last 5 years and what has its impact been on the industry? For any questions related to Acefylline Piperazine Market in United States, Acefylline Piperazine Market in United States Size, Acefylline Piperazine Market in United States Growth, Acefylline Piperazine Market in United States Analysis, Acefylline Piperazine Market in United States Report, Acefylline Piperazine Market in United States Share, Acefylline Piperazine Market in United States Trends, Acefylline Piperazine Market in United States Forecast, Acefylline Piperazine Companies, write Lucintel analyst at email: helpdesk@lucintel.com. We will be glad to get back to you soon.